The effects of aging on neuropil structure in mouse somatosensory cortex-A 3D electron microscopy analysis of layer 1
- PMID: 29966021
- PMCID: PMC6028106
- DOI: 10.1371/journal.pone.0198131
The effects of aging on neuropil structure in mouse somatosensory cortex-A 3D electron microscopy analysis of layer 1
Abstract
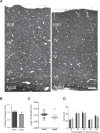
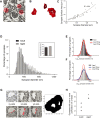
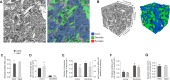
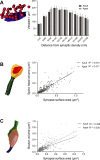
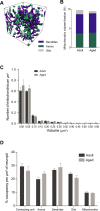
This study has used dense reconstructions from serial EM images to compare the neuropil ultrastructure and connectivity of aged and adult mice. The analysis used models of axons, dendrites, and their synaptic connections, reconstructed from volumes of neuropil imaged in layer 1 of the somatosensory cortex. This shows the changes to neuropil structure that accompany a general loss of synapses in a well-defined brain region. The loss of excitatory synapses was balanced by an increase in their size such that the total amount of synaptic surface, per unit length of axon, and per unit volume of neuropil, stayed the same. There was also a greater reduction of inhibitory synapses than excitatory, particularly those found on dendritic spines, resulting in an increase in the excitatory/inhibitory balance. The close correlations, that exist in young and adult neurons, between spine volume, bouton volume, synaptic size, and docked vesicle numbers are all preserved during aging. These comparisons display features that indicate a reduced plasticity of cortical circuits, with fewer, more transient, connections, but nevertheless an enhancement of the remaining connectivity that compensates for a generalized synapse loss.
Conflict of interest statement
The authors would like to declare the following competing interest: this study was funded in part by a grant from the Novartis Biomedical Foundation. This does not alter our adherence to PLOS ONE policies on sharing data and materials.
Figures

References
-
- Hof PR, Morrison JH (2004) The aging brain: morphomolecular senescence of cortical circuits. Trends Neurosci 27:607–613 doi: 10.1016/j.tins.2004.07.013 - DOI - PubMed
-
- Morrison JH, Baxter MG (2012) The ageing cortical synapse: hallmarks and implications for cognitive decline. Nat Rev Neurosci 13:240–250 doi: 10.1038/nrn3200 - DOI - PMC - PubMed
-
- Burke S.N., and Barnes C.A. (2006). Neural plasticity in the ageing brain. Nat. Rev. Neurosci. 7, 30–40. doi: 10.1038/nrn1809 - DOI - PubMed
-
- Dickstein DL, Weaver CM, Luebke JI, Hof PR (2013) Dendritic spine changes associated with normal aging. Neuroscience 251:21–32 doi: 10.1016/j.neuroscience.2012.09.077 - DOI - PMC - PubMed
-
- Peters A, Sethares C, Moss MB (1998) The effects of aging on layer 1 in area 46 of prefrontal cortex in the rhesus monkey. Cereb Cortex 8:671–684 - PubMed
Publication types
MeSH terms
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

