Patients and investigators prefer measures of absolute risk in subgroups for pragmatic randomized trials
- PMID: 29966732
- PMCID: PMC6175611
- DOI: 10.1016/j.jclinepi.2018.06.009
Patients and investigators prefer measures of absolute risk in subgroups for pragmatic randomized trials
Abstract
Objectives: Pragmatic randomized trials are important tools for shared decision-making, but no guidance exists on patients' preferences for types of causal information. We aimed to assess preferences of patients and investigators toward causal effects in pragmatic randomized trials.
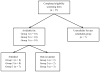
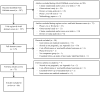
Study design and setting: We (a) held three focus groups with patients (n = 23) in Boston, MA; (b) surveyed (n = 12) and interviewed (n = 5) investigators with experience conducting pragmatic trials; and (c) conducted a systematic literature review of pragmatic trials (n = 63).
Results: Patients were distrustful of new-to-market medications unless substantially more effective than existing choices, preferred stratified absolute risks, and valued adherence-adjusted analyses when they expected to adhere. Investigators wanted both intention-to-treat and per-protocol effects but felt methods for estimating per-protocol effects were lacking. When estimating per-protocol effects, many pragmatic trials used inappropriate methods to adjust for adherence and loss to follow-up.
Conclusion: We made four recommendations for pragmatic trials to improve patient centeredness: (1) focus on superiority in effectiveness or safety, rather than noninferiority; (2) involve patients in specifying a priori subgroups; (3) report absolute measures of risk; and (4) complement intention-to-treat effect estimates with valid per-protocol effect estimates.
Keywords: Adherence adjustment; Causal inference; Health communication; Intention to treat; Patient preferences; Per protocol; Pragmatic trial.
Copyright © 2018 Elsevier Inc. All rights reserved.
Figures
References
-
- Härter M, Simon D. Shared decision-making in diverse health care systems—Translating research into practice. Patient Education and Counseling. 2008;73(3):399–401. - PubMed
-
- Joosten EAG, DeFuentes-Merillas L, de Weert GH, Sensky T, van der Staak CPF, de Jong CAJ. Systematic Review of the Effects of Shared Decision-Making on Patient Satisfaction, Treatment Adherence and Health Status. Psychotherapy and psychosomatics. 2008;77(4):219–26. - PubMed
-
- Zipkin DA, Umscheid CA, Keating NL, Allen E, Aung K, Beyth R, et al. Evidence-based risk communication: a systematic review. Ann Intern Med. 2014;161(4):270–80. - PubMed
-
- Epstein AM, Taylor WC, Seage GR., 3rd Effects of patients’ socioeconomic status and physicians’ training and practice on patient-doctor communication. Am J Med. 1985;78(1):101–6. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical