Engineering glycoside hydrolase stability by the introduction of zinc binding
- PMID: 29968680
- PMCID: PMC6038386
- DOI: 10.1107/S2059798318006678
Engineering glycoside hydrolase stability by the introduction of zinc binding
Abstract
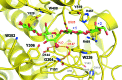
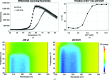
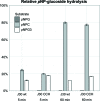
The development of robust enzymes, in particular cellulases, is a key step in the success of biological routes to `second-generation' biofuels. The typical sources of the enzymes used to degrade biomass include mesophilic and thermophilic organisms. The endoglucanase J30 from glycoside hydrolase family 9 was originally identified through metagenomic analyses of compost-derived bacterial consortia. These studies, which were tailored to favor growth on targeted feedstocks, have already been shown to identify cellulases with considerable thermal tolerance. The amino-acid sequence of J30 shows comparably low identity to those of previously analyzed enzymes. As an enzyme that combines a well measurable activity with a relatively low optimal temperature (50°C) and a modest thermal tolerance, it offers the potential for structural optimization aimed at increased stability. Here, the crystal structure of wild-type J30 is presented along with that of a designed triple-mutant variant with improved characteristics for industrial applications. Through the introduction of a structural Zn2+ site, the thermal tolerance was increased by more than 10°C and was paralleled by an increase in the catalytic optimum temperature by more than 5°C.
Keywords: X-ray crystallography; glycoside hydrolases; protein engineering; thermal stability.
open access.
Figures

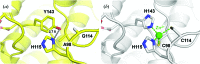
Similar articles
-
Glycoside hydrolase activities of thermophilic bacterial consortia adapted to switchgrass.Appl Environ Microbiol. 2011 Aug 15;77(16):5804-12. doi: 10.1128/AEM.00032-11. Epub 2011 Jul 1. Appl Environ Microbiol. 2011. PMID: 21724886 Free PMC article.
-
Comparison of family 12 glycoside hydrolases and recruited substitutions important for thermal stability.Protein Sci. 2003 Apr;12(4):848-60. doi: 10.1110/ps.0237703. Protein Sci. 2003. PMID: 12649442 Free PMC article.
-
Engineering Robust Cellulases for Tailored Lignocellulosic Degradation Cocktails.Int J Mol Sci. 2020 Feb 26;21(5):1589. doi: 10.3390/ijms21051589. Int J Mol Sci. 2020. PMID: 32111065 Free PMC article. Review.
-
Dynamic Perturbation of the Active Site Determines Reversible Thermal Inactivation in Glycoside Hydrolase Family 12.J Chem Inf Model. 2017 Feb 27;57(2):288-297. doi: 10.1021/acs.jcim.6b00692. Epub 2017 Feb 9. J Chem Inf Model. 2017. PMID: 28145703
-
Characteristics, protein engineering and applications of microbial thermostable pullulanases and pullulan hydrolases.Appl Microbiol Biotechnol. 2016 Jul;100(13):5661-79. doi: 10.1007/s00253-016-7572-y. Epub 2016 May 3. Appl Microbiol Biotechnol. 2016. PMID: 27142298 Review.
Cited by
-
New thermostable endoglucanase from Spirochaeta thermophila and its mutants with altered substrate preferences.Appl Microbiol Biotechnol. 2021 Feb;105(3):1133-1145. doi: 10.1007/s00253-020-11077-x. Epub 2021 Jan 11. Appl Microbiol Biotechnol. 2021. PMID: 33427929
References
-
- Adams, P. D. et al. (2010). Acta Cryst. D66, 213–221. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

