Hexane-Isopropanolic Extract of Tungrymbai, a North-East Indian fermented soybean food prevents hepatic steatosis via regulating AMPK-mediated SREBP/FAS/ACC/HMGCR and PPARα/CPT1A/UCP2 pathways
- PMID: 29968750
- PMCID: PMC6030226
- DOI: 10.1038/s41598-018-27607-7
Hexane-Isopropanolic Extract of Tungrymbai, a North-East Indian fermented soybean food prevents hepatic steatosis via regulating AMPK-mediated SREBP/FAS/ACC/HMGCR and PPARα/CPT1A/UCP2 pathways
Abstract
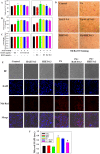
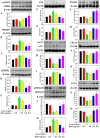
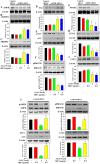
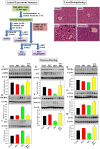
This study for the first time examined the prophylactic role of Tungrymbai, a well-known fermented soybean food of North-East India, against hepatic steatosis. Treatment with hexane-isopropanolic (2:1, HIET) but not hydro-alcoholic (70% ethanol, HAET) extract dose-dependently (0.1, 0.2, or 0.3 µg/mL) reduced the intracellular lipid accumulation as shown by lower triglyceride levels and both Oil Red O and Nile Red staining in palmitate (PA, 0.75 mM)-treated hepatocytes. Immunobloting, mRNA expression, and knock-down studies demonstrated the role of AMPK-mediated SREBP/FAS/ACC/HMGCR and PPARα/CPT1A/UCP2 signaling pathways in facilitating the beneficial role of HIET against lipid accumulation in PA-treated hepatocytes. Animal studies further showed a positive effect of HIET (20 µg/kg BW, 8 weeks, daily) in regulating AMPK/SREBP/PPARα signaling pathways and reducing body weight gain, plasma lipid levels, and hepatic steatosis in high fat diet (HFD)-fed mice. Histological analyses also revealed the beneficial effect of HIET in reducing hepatic fat accumulation in HFD mice. Chemical profiling (HRMS, IR, and HPLC) demonstrated the presence of menaquinone-7 (vitamin K2) as one of the bio-active principle(s) in HIET. Combining all, this study demonstrates the positive effect of HIET on reducing hepatic steatosis via regulating AMPK/SREBP/PPARα signaling pathway.
Conflict of interest statement
The authors declare no competing interests.
Figures
Similar articles
-
Alisol A 24-Acetate Prevents Hepatic Steatosis and Metabolic Disorders in HepG2 Cells.Cell Physiol Biochem. 2016;40(3-4):453-464. doi: 10.1159/000452560. Epub 2016 Nov 25. Cell Physiol Biochem. 2016. PMID: 27889747
-
Dietary α-lactalbumin induced fatty liver by enhancing nuclear liver X receptor αβ/sterol regulatory element-binding protein-1c/PPARγ expression and minimising PPARα/carnitine palmitoyltransferase-1 expression and AMP-activated protein kinase α phosphorylation associated with atherogenic dyslipidaemia, insulin resistance and oxidative stress in Balb/c mice.Br J Nutr. 2017 Dec;118(11):914-929. doi: 10.1017/S000711451700232X. Epub 2017 Nov 27. Br J Nutr. 2017. PMID: 29173234
-
Honokiol activates the LKB1-AMPK signaling pathway and attenuates the lipid accumulation in hepatocytes.Toxicol Appl Pharmacol. 2015 Apr 15;284(2):113-24. doi: 10.1016/j.taap.2015.02.020. Epub 2015 Feb 28. Toxicol Appl Pharmacol. 2015. PMID: 25737164
-
Purple sweet potato anthocyanins attenuate hepatic lipid accumulation through activating adenosine monophosphate-activated protein kinase in human HepG2 cells and obese mice.Nutr Res. 2011 Dec;31(12):896-906. doi: 10.1016/j.nutres.2011.09.026. Nutr Res. 2011. PMID: 22153515
-
Chlorogenic acid improves food allergy through the AMPK/ACC/CPT-1 pathway.J Food Biochem. 2022 Dec;46(12):e14505. doi: 10.1111/jfbc.14505. Epub 2022 Dec 1. J Food Biochem. 2022. PMID: 36455237 Review.
Cited by
-
Berberine Improves Glucose and Lipid Metabolism in HepG2 Cells Through AMPKα1 Activation.Front Pharmacol. 2020 May 8;11:647. doi: 10.3389/fphar.2020.00647. eCollection 2020. Front Pharmacol. 2020. PMID: 32457629 Free PMC article.
-
Bile acids supplementation modulates lipid metabolism, intestinal function, and cecal microbiota in geese.Front Microbiol. 2023 May 26;14:1185218. doi: 10.3389/fmicb.2023.1185218. eCollection 2023. Front Microbiol. 2023. PMID: 37303790 Free PMC article.
-
Virgin Camellia Seed Oil Improves Glycolipid Metabolism in the Kidney of High Fat-Fed Rats through AMPK-SREBP Pathway.Nutrients. 2023 Nov 23;15(23):4888. doi: 10.3390/nu15234888. Nutrients. 2023. PMID: 38068746 Free PMC article.
-
The novel long noncoding RNA Lnc19959.2 modulates triglyceride metabolism-associated genes through the interaction with Purb and hnRNPA2B1.Mol Metab. 2020 Jul;37:100996. doi: 10.1016/j.molmet.2020.100996. Epub 2020 Apr 14. Mol Metab. 2020. PMID: 32302712 Free PMC article.
-
9-HODE and 9-HOTrE alter mitochondrial metabolism, increase triglycerides, and perturb fatty acid uptake and synthesis associated gene expression in HepG2 cells.Prostaglandins Leukot Essent Fatty Acids. 2024 Mar;202:102635. doi: 10.1016/j.plefa.2024.102635. Epub 2024 Aug 13. Prostaglandins Leukot Essent Fatty Acids. 2024. PMID: 39142221 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

