Stereospecific Radical-Mediated B12-Dependent Methyl Transfer by the Fosfomycin Biosynthesis Enzyme Fom3
- PMID: 29969250
- PMCID: PMC6103835
- DOI: 10.1021/acs.biochem.8b00616
Stereospecific Radical-Mediated B12-Dependent Methyl Transfer by the Fosfomycin Biosynthesis Enzyme Fom3
Abstract
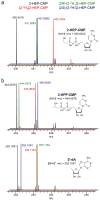
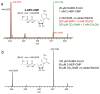
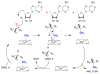
Fom3, the antepenultimate enzyme in the fosfomycin biosynthetic pathway in Streptomyces spp., is a class B cobalamin-dependent radical SAM methyltransferase that catalyzes methylation of (5'-cytidylyl)-2-hydroxyethylphosphonate (2-HEP-CMP) to form (5'-cytidylyl)-2-hydroxypropylphosphonate (2-HPP-CMP). Previously, the reaction of Fom3 with 2-HEP-CMP produced 2-HPP-CMP with mixed stereochemistry at C2. Mechanistic characterization has been challenging because of insoluble expression and poor cobalamin (B12) incorporation in Escherichia coli. Recently, soluble E. coli expression and incorporation of cobalamin into Fom3 were achieved by overexpression of the BtuCEDFB cobalamin uptake system. Herein, we use this new method to obtain Fom3 from Streptomyces wedmorensis. We show that the initiator 5'-deoxyadenosyl radical stereospecifically abstracts the pro- R hydrogen atom from the C2 position of 2-HEP-CMP and use the downstream enzymes FomD and Fom4 to demonstrate that our preparation of Fom3 produces only (2 S)-2-HPP-CMP. Additionally, we show that the added methyl group originates from SAM under multiple-turnover conditions, but the first turnover uses a methyl donor already present on the enzyme; furthermore, cobalamin isolated from Fom3 reaction mixtures contains methyl groups derived from SAM. These results are consistent with a model in which Fom3 catalyzes methyl transfer from SAM to cobalamin and the resulting methylcobalamin (MeCbl) is the ultimate methyl source for the reaction.
Conflict of interest statement
The authors declare no competing financial interest.
Figures


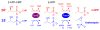
Similar articles
-
Stereochemical and Mechanistic Investigation of the Reaction Catalyzed by Fom3 from Streptomyces fradiae, a Cobalamin-Dependent Radical S-Adenosylmethionine Methylase.Biochemistry. 2018 Aug 21;57(33):4972-4984. doi: 10.1021/acs.biochem.8b00693. Epub 2018 Aug 9. Biochemistry. 2018. PMID: 30036047 Free PMC article.
-
C-Methylation Catalyzed by Fom3, a Cobalamin-Dependent Radical S-adenosyl-l-methionine Enzyme in Fosfomycin Biosynthesis, Proceeds with Inversion of Configuration.Biochemistry. 2018 Aug 21;57(33):4963-4966. doi: 10.1021/acs.biochem.8b00614. Epub 2018 Jul 12. Biochemistry. 2018. PMID: 29966085
-
Methylcobalamin-Dependent Radical SAM C-Methyltransferase Fom3 Recognizes Cytidylyl-2-hydroxyethylphosphonate and Catalyzes the Nonstereoselective C-Methylation in Fosfomycin Biosynthesis.Biochemistry. 2017 Jul 18;56(28):3519-3522. doi: 10.1021/acs.biochem.7b00472. Epub 2017 Jul 7. Biochemistry. 2017. PMID: 28678474
-
Radical-mediated enzymatic methylation: a tale of two SAMS.Acc Chem Res. 2012 Apr 17;45(4):555-64. doi: 10.1021/ar200202c. Epub 2011 Nov 18. Acc Chem Res. 2012. PMID: 22097883 Free PMC article. Review.
-
Mechanistic diversity of radical S-adenosylmethionine (SAM)-dependent methylation.J Biol Chem. 2015 Feb 13;290(7):3995-4002. doi: 10.1074/jbc.R114.607044. Epub 2014 Dec 4. J Biol Chem. 2015. PMID: 25477520 Free PMC article. Review.
Cited by
-
Biosynthesis of macrocyclic peptides with C-terminal β-amino-α-keto acid groups by three different metalloenzymes.bioRxiv [Preprint]. 2024 Jan 17:2023.10.30.564719. doi: 10.1101/2023.10.30.564719. bioRxiv. 2024. Update in: ACS Cent Sci. 2024 Apr 11;10(5):1022-1032. doi: 10.1021/acscentsci.4c00088. PMID: 37965205 Free PMC article. Updated. Preprint.
-
Biosynthesis of Macrocyclic Peptides with C-Terminal β-Amino-α-keto Acid Groups by Three Different Metalloenzymes.ACS Cent Sci. 2024 Apr 11;10(5):1022-1032. doi: 10.1021/acscentsci.4c00088. eCollection 2024 May 22. ACS Cent Sci. 2024. PMID: 38799663 Free PMC article.
-
Studies of GenK and OxsB, two B12-dependent radical SAM enzymes involved in natural product biosynthesis.Methods Enzymol. 2022;669:71-90. doi: 10.1016/bs.mie.2021.12.014. Epub 2022 Feb 3. Methods Enzymol. 2022. PMID: 35644181 Free PMC article.
-
The Hydrogenobyric Acid Structure Reveals the Corrin Ligand as an Entatic State Module Empowering B12 Cofactors for Catalysis.Angew Chem Int Ed Engl. 2019 Jul 29;58(31):10756-10760. doi: 10.1002/anie.201904713. Epub 2019 Jun 26. Angew Chem Int Ed Engl. 2019. PMID: 31115943 Free PMC article.
-
Enzymatic assembly of carbon-carbon bonds via iron-catalysed sp3 C-H functionalization.Nature. 2019 Jan;565(7737):67-72. doi: 10.1038/s41586-018-0808-5. Epub 2018 Dec 19. Nature. 2019. PMID: 30568304 Free PMC article.
References
-
- Kahan FM, Kahan JS, Cassidy PJ, Kropp H. The mechanism of action of fosfomycin (phosphonomycin) Ann N Y Acad Sci. 1974;235:364–386. - PubMed
-
- Marquardt JL, Brown ED, Lane WS, Haley TM, Ichikawa Y, Wong CH, Walsh CT. Kinetics, stoichiometry, and identification of the reactive thiolate in the inactivation of UDP-GlcNAc enolpyruvoyl transferase by the antibiotic fosfomycin. Biochemistry. 1994;33:10646–10651. - PubMed
-
- Stein GE. Single-dose treatment of acute cystitis with fosfomycin tromethamine. Ann Pharmacother. 1998;32:215–219. - PubMed
-
- Bailey RR. Management of lower urinary tract infections. Drugs. 1993;45(Suppl 3):139–144. - PubMed
-
- Seto H, Kuzuyama T. Bioactive Natural Products with Carbon-Phosphorus Bonds and Their Biosynthesis. Nat Prod Rep. 1999;16:589–596. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

