An in vitro and in vivo study on the properties of hollow polycaprolactone cell-delivery particles
- PMID: 29969443
- PMCID: PMC6029779
- DOI: 10.1371/journal.pone.0198248
An in vitro and in vivo study on the properties of hollow polycaprolactone cell-delivery particles
Abstract
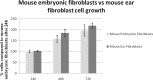
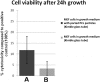
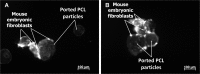
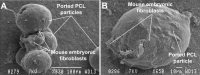
The field of dermal fillers is evolving rapidly and numerous products are currently on the market. Biodegradable polymers such as polycaprolactone (PCL) have been found to be compatible with several body tissues, and this makes them an ideal material for dermal filling purposes. Hollow PCL spheres were developed by the Council for Scientific and Industrial Research (CSIR) to serve both as an anchor point and a "tissue harbour" for cells. Particles were tested for cytotoxicity and cell adherence using mouse embryo fibroblasts (MEF). MEFs adhered to the particles and no significant toxic effects were observed based on morphology, cell growth, cell viability and cell cycle analysis, suggesting that the particles are suitable candidates for cell delivery systems in an in vivo setting. The objective of providing a "tissue harbour" was however not realized, as cells did not preferentially migrate into the ported particles. In vivo studies were conducted in BALB/c mice into whom particles were introduced at the level of the hypodermis. Mice injected with PCL particles (ported and non-ported; with or without MEFs) showed evidence of local inflammation and increased adipogenesis at the site of injection, as well as a systemic inflammatory response. These effects were also observed in mice that received apparently inert (polystyrene) particles. Ported PCL particles can therefore act as a cell delivery system and through their ability to induce adipogenesis, may also serve as a dermal bulking agent.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures





References
-
- Kulichova D, Borovaya A, Ruzicka T, Thomas P, Gauglitz GG. Understanding the safety and tolerability of facial filling therapeutics. Expert opinion on drug safety. 2014:1–12. Epub 2014/07/19. doi: 10.1517/14740338.2014.939168 . - DOI - PubMed
-
- Funt D, Pavicic T. Dermal fillers in aesthetics: an overview of adverse events and treatment approaches. Clinical, cosmetic and investigational dermatology. 2013;6:295–316. Epub 2013/12/24. doi: 10.2147/CCID.S50546 ; PubMed Central PMCID: PMCPmc3865975. - DOI - PMC - PubMed
-
- ASPS. 2012 Plastic Surgery Procedural Statistics2012 15/07/2014. Available from: http://www.plasticsurgery.org/Documents/news-resources/statistics/2012-P....
-
- Gilbert E, Calvisi L. Midface and perioral volume restoration: a conversation between the US and Italy. Journal of drugs in dermatology: JDD. 2014;13(1):67–74. Epub 2014/01/05. . - PubMed
-
- De Boulle K. Management of complications after implantation of fillers. J Cosmet Dermatol. 2004;3(1):2–15. Epub 2006/12/14. JCD058 [pii] doi: 10.1111/j.1473-2130.2004.00058.x . - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

