The duration of hypothermia affects short-term neuroprotection in a mouse model of neonatal hypoxic ischaemic injury
- PMID: 29969470
- PMCID: PMC6029790
- DOI: 10.1371/journal.pone.0199890
The duration of hypothermia affects short-term neuroprotection in a mouse model of neonatal hypoxic ischaemic injury
Abstract
Neonatal hypoxic-ischaemic encephalopathy (HIE) is major cause of neonatal mortality and morbidity. Therapeutic hypothermia is standard clinical care for moderate hypoxic-ischaemic (HI) brain injury, however it reduces the risk of death and disability only by 11% and 40% of the treated infants still develop disabilities. Thus it is necessary to develop supplementary therapies to complement therapeutic hypothermia in the treatment of neonatal HIE. The modified Rice-Vannucci model of HI in the neonatal mouse is well developed and widely applied with different periods of hypothermia used as neuroprotective strategy in combination with other agents. However, different studies use different periods, time of initiation and duration of hypothermia following HI, with subsequent varying degrees of neuroprotection. So far most rodent data is obtained using exposure to 5-6h of therapeutic hypothermia. Our aim was to compare the effect of exposure to three different short periods of hypothermia (1h, 1.5h and 2h) following HI insult in the postnatal day 7 C57/Bl6 mouse, and to determine the shortest period providing neuroprotection. Our data suggests that 1h and 1.5h of hypothermia delayed by 20min following a 60min exposure to 8%O2 do not prove neuroprotective. However, 2h of hypothermia significantly reduced tissue loss, TUNEL+ cell death and microglia and astroglia activation. We also observed improved functional outcome 7 days after HI. We suggest that the minimal period of cooling necessary to provide moderate short term neuroprotection and appropriate for the development and testing of combined treatment is 2h.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures
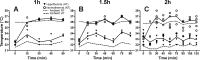
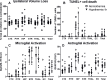
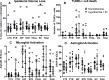


Similar articles
-
Neuroprotection with hypothermia and allopurinol in an animal model of hypoxic-ischemic injury: Is it a gender question?PLoS One. 2017 Sep 20;12(9):e0184643. doi: 10.1371/journal.pone.0184643. eCollection 2017. PLoS One. 2017. PMID: 28931035 Free PMC article.
-
Neuroprotective exendin-4 enhances hypothermia therapy in a model of hypoxic-ischaemic encephalopathy.Brain. 2018 Oct 1;141(10):2925-2942. doi: 10.1093/brain/awy220. Brain. 2018. PMID: 30165597 Free PMC article.
-
Endogenous hypothermic response to hypoxia reduces brain injury: Implications for modeling hypoxic-ischemic encephalopathy and therapeutic hypothermia in neonatal mice.Exp Neurol. 2016 Sep;283(Pt A):264-75. doi: 10.1016/j.expneurol.2016.06.024. Epub 2016 Jun 25. Exp Neurol. 2016. PMID: 27349408
-
Neuroprotection for neonatal hypoxic-ischemic encephalopathy: A review of novel therapies evaluated in clinical studies.Dev Med Child Neurol. 2025 May;67(5):591-599. doi: 10.1111/dmcn.16184. Epub 2024 Nov 20. Dev Med Child Neurol. 2025. PMID: 39563426 Free PMC article. Review.
-
Targeting the Multiple Complex Processes of Hypoxia-Ischemia to Achieve Neuroprotection.Int J Mol Sci. 2024 May 17;25(10):5449. doi: 10.3390/ijms25105449. Int J Mol Sci. 2024. PMID: 38791487 Free PMC article. Review.
Cited by
-
Ambient Temperature Is Correlated With the Severity of Neonatal Hypoxic-Ischemic Brain Injury via Microglial Accumulation in Mice.Front Pediatr. 2022 May 6;10:883556. doi: 10.3389/fped.2022.883556. eCollection 2022. Front Pediatr. 2022. PMID: 35601427 Free PMC article.
-
Preventing Brain Damage from Hypoxic-Ischemic Encephalopathy in Neonates: Update on Mesenchymal Stromal Cells and Umbilical Cord Blood Cells.Am J Perinatol. 2022 Dec;39(16):1754-1763. doi: 10.1055/s-0041-1726451. Epub 2021 Apr 14. Am J Perinatol. 2022. PMID: 33853147 Free PMC article. Review.
-
Mesenchymal Stromal Cell Derived Extracellular Vesicles Reduce Hypoxia-Ischaemia Induced Perinatal Brain Injury.Front Physiol. 2019 Mar 19;10:282. doi: 10.3389/fphys.2019.00282. eCollection 2019. Front Physiol. 2019. PMID: 30941062 Free PMC article.
-
Treatment and new progress of neonatal hypoxic-ischemic brain damage.Histol Histopathol. 2020 Sep;35(9):929-936. doi: 10.14670/HH-18-214. Epub 2020 Mar 13. Histol Histopathol. 2020. PMID: 32167570 Review.
-
S100a8 silencing attenuates inflammation, oxidative stress and apoptosis in BV2 cells induced by oxygen‑glucose deprivation and reoxygenation by upregulating GAB1 expression.Mol Med Rep. 2021 Jan;23(1):64. doi: 10.3892/mmr.2020.11702. Epub 2020 Nov 20. Mol Med Rep. 2021. PMID: 33215218 Free PMC article.
References
-
- Kurinczuk JJ, White-Koning M, Badawi N. Epidemiology of neonatal encephalopathy and hypoxic-ischaemic encephalopathy. Early Human Development. 2010. pp. 329–338. doi: 10.1016/j.earlhumdev.2010.05.010 - DOI - PubMed
-
- Lawn JE, Cousens S, Zupan J. Neonatal Survival 1 4 million neonatal deaths: When? Where? Why? 2005; 891–900. - PubMed
-
- Lawn JE, Kerber K, Enweronu-Laryea C, Cousens S. 3.6 Million Neonatal Deaths-What Is Progressing and What Is Not? Semin Perinatol. Elsevier Inc.; 2010;34: 371–386. doi: 10.1053/j.semperi.2010.09.011 - DOI - PubMed
-
- Kattwinkel J, Perlman J, Aziz K, Colby C, Fairchild K, Gallagher J, et al. Part 15: Neonatal resuscitation: 2010 American Heart Association guidelines for cardiopulmonary resuscitation and emergency cardiovascular care. Circ J Am Hear Assoc. 2010;122: S909–S919. doi: 10.1161/CIRCULATIONAHA.110.971119 - DOI - PubMed
-
- Marion D, Bullock MR. Current and Future Role of Therapeutic Hypothermia. Mary Ann Liebert, Inc. 140 Huguenot Street, 3rd Floor New Rochelle, NY 10801 USA; 2009;
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

