Screening for biomarkers reflecting the progression of Babesia microti infection
- PMID: 29970143
- PMCID: PMC6029176
- DOI: 10.1186/s13071-018-2951-0
Screening for biomarkers reflecting the progression of Babesia microti infection
Abstract
Background: Babesiosis is caused by the invasion of erythrocytes by parasites of the Babesia spp. Babesia microti is one of the primary causative agents of human babesiosis. To better understand the status of the disease, discovering key biomarkers of the different infection stages is crucial.
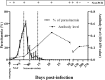
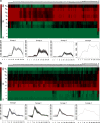
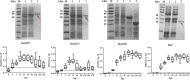
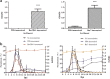
Results: This study investigated B. microti infection in the mouse model from 0 to 270 days post-infection (dpi), using blood smears, PCR assays and ELISA. PCR assays showed a higher sensitivity when compared to microscopic examination. Specific IgG antibodies could be detected from 7 days to 270 dpi. Two-dimensional electrophoresis was combined with western blotting and mass spectrometric analysis to screen for specific reactive antigens during both the peak parasitaemia period (7 dpi) and IgG antibody response peak period (30 dpi) by the infected mice plasma. The 87 positive reactive proteins were identified and then expressed with the wheat germ cell-free system. Protein microarrays of all 87 targeted proteins were produced and hybridized with the serial plasma of infected mice model. Based on the antigen reaction profile during the infection procedure, 6 antigens were selected and expressed in Escherichia coli. Due to an early response to IgM, lower immunoreactivity levels of IgG after two months and higher immunoreactivity level IgG during nine months, four recombinant proteins were selected for further characterization, namely rBm2D97(CCF75281.1), rBm2D33(CCF74637.1), rBm2D41(CCF75408.1) and rBm7(CCF73510.1). The diagnostic efficacy of the four recombinant protein candidates was evaluated in a clinical setting using babesiosis patient plasma. The rBm2D33 showed the highest sensitivity with a positive rate of 62.5%. Additional characterization of the two candidate proteins using a mouse vaccination assay, demonstrated that rBm2D41 could reduce peak parasitaemia by 37.4%, indicating its efficacy in preventing severe babesiosis.
Conclusions: The detection technologies of microscopic examination, PCR assays and antibody tests showed different sensitivities and accuracy during the different stages of B. microti infection. Antibody detection has a unique significance for B. microti infection in the asymptomatic stages. Using immunoreactivity profiles, biomarkers for disease progression were identified and represent useful information for future the diagnosis and vaccine development for this serious disease of public health significance.
Keywords: Babesia microti; Cell free expression system; Expression and evaluation of proteins; Mice model; Protein microarray; Screening biomarker; Strain ATCC®PRA-99TM.
Conflict of interest statement
Ethics approval and consent to participate
Ethical clearance for the collection and detection of human samples was obtained from the Ethics Committee of the National Institute of Parasitic Diseases (NIPD), Chinese Center for Disease Control and Prevention (China CDC). The objectives, procedures and potential risks were verbally explained to all participants. Signed written informed consent was obtained from all study participants. All participants were adults in this study. Animals were handled in accordance with good animal practice strictly according to the Animal Ethics Procedures and Guidelines of the People’s Republic of China. The protocol for sampling from animals had been approved by the Animal Welfare& Ethics Committee of the National Institute of Parasitic Diseases, Chinese Center for Disease Control and Prevention in Shanghai (Permit No: IPD-2012-5).
Consent for publication
We have obtained consent to publish from the participants (or legal parents or guardians for children) to report individual patient data.
Competing interests
The authors declare that they have no competing interests.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Figures



Similar articles
-
Molecular characterization of Babesia microti seroreactive antigen 5-1-1 and development of rapid detection methods for anti-B. microti antibodies in serum.Acta Trop. 2018 Sep;185:371-379. doi: 10.1016/j.actatropica.2018.03.020. Epub 2018 Mar 17. Acta Trop. 2018. PMID: 29559329
-
Identification of Babesia microti-specific immunodominant epitopes and development of a peptide EIA for detection of antibodies in serum.Transfusion. 2002 Nov;42(11):1488-96. doi: 10.1046/j.1537-2995.2002.00215.x. Transfusion. 2002. PMID: 12421223
-
Babesia microti Protein BmSP44 Is a Novel Protective Antigen in a Mouse Model of Babesiosis.Front Immunol. 2020 Jul 7;11:1437. doi: 10.3389/fimmu.2020.01437. eCollection 2020. Front Immunol. 2020. PMID: 32733477 Free PMC article.
-
Vaccination against babesiosis using recombinant GPI-anchored proteins.Int J Parasitol. 2019 Feb;49(2):175-181. doi: 10.1016/j.ijpara.2018.12.002. Epub 2019 Jan 24. Int J Parasitol. 2019. PMID: 30684517 Review.
-
[Babesosis--difficulty of diagnosis].Wiad Parazytol. 2001;47(3):527-33. Wiad Parazytol. 2001. PMID: 16894770 Review. Polish.
Cited by
-
A Fluorescent Recombinase Aided Amplification Assay for Detection of Babesia microti.Korean J Parasitol. 2022 Jun;60(3):201-205. doi: 10.3347/kjp.2022.60.3.201. Epub 2022 Jun 30. Korean J Parasitol. 2022. PMID: 35772739 Free PMC article.
-
Advances in Babesia Vaccine Development: An Overview.Pathogens. 2023 Feb 11;12(2):300. doi: 10.3390/pathogens12020300. Pathogens. 2023. PMID: 36839572 Free PMC article. Review.
-
Exploration of Serum Marker Proteins in Mice Induced by Babesia microti Infection Using a Quantitative Proteomic Approach.Protein J. 2021 Feb;40(1):119-130. doi: 10.1007/s10930-020-09952-7. Epub 2021 Jan 2. Protein J. 2021. PMID: 33387247
-
Babesia microti: Pathogen Genomics, Genetic Variability, Immunodominant Antigens, and Pathogenesis.Front Microbiol. 2021 Sep 3;12:697669. doi: 10.3389/fmicb.2021.697669. eCollection 2021. Front Microbiol. 2021. PMID: 34539601 Free PMC article. Review.
-
A conserved protein of Babesia microti elicits partial protection against Babesia and Plasmodium infection.Parasit Vectors. 2023 Aug 30;16(1):306. doi: 10.1186/s13071-023-05825-x. Parasit Vectors. 2023. PMID: 37649042 Free PMC article.
References
-
- Fox LAM, Wingerter S, Ahmed A, Arnold A, Chou J, Rhein L, et al. Neonatal babesiosis - case report and review of the literature. Pediatr Infect Dis J. 2006;25:169–73. - PubMed
-
- Levine ND. The protozoan phylum Apicomplexa. Volumes I and II. Boca Raton: CRC Press; 1988.
MeSH terms
Substances
Grants and funding
- GWIV-29/the Fourth Round of a Three-Year Public Health Action Plan (2015-2017) in Shanghai
- GW-11/Strengthen Action Plan for Shanghai Public Health System Construction 2011-2013
- 2009ZX10004-302/National S and T Major Program
- 201502021, 201202019/Chinese Special Program for Scientific Research of Public Health
- 2014DFA31130/International Science and Technology Cooperation Program of China
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

