Effects of dapagliflozin and n-3 carboxylic acids on non-alcoholic fatty liver disease in people with type 2 diabetes: a double-blind randomised placebo-controlled study
- PMID: 29971527
- PMCID: PMC6096619
- DOI: 10.1007/s00125-018-4675-2
Effects of dapagliflozin and n-3 carboxylic acids on non-alcoholic fatty liver disease in people with type 2 diabetes: a double-blind randomised placebo-controlled study
Abstract
Aims/hypothesis: The EFFECT-II study aimed to investigate the effects of dapagliflozin and omega-3 (n-3) carboxylic acids (OM-3CA), individually or combined, on liver fat content in individuals with type 2 diabetes and non-alcoholic fatty liver disease (NAFLD).
Methods: This randomised placebo-controlled double-blind parallel-group study was performed at five clinical research centres at university hospitals in Sweden. 84 participants with type 2 diabetes and NAFLD were randomly assigned 1:1:1:1 to four treatments by a centralised randomisation system, and all participants as well as investigators and staff involved in the study conduct and analyses were blinded to treatments. Each group received oral doses of one of the following: 10 mg dapagliflozin (n = 21), 4 g OM-3CA (n = 20), a combination of both (n = 22) or placebo (n = 21). The primary endpoint was liver fat content assessed by MRI (proton density fat fraction [PDFF]) and, in addition, total liver volume and markers of glucose and lipid metabolism as well as of hepatocyte injury and oxidative stress were assessed at baseline and after 12 weeks of treatment (completion of the trial).
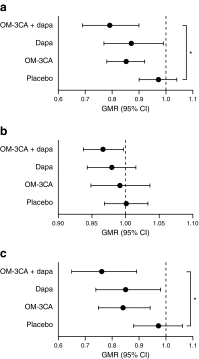
Results: Participants had a mean age of 65.5 years (SD 5.9), BMI 31.2 kg/m2 (3.5) and liver PDFF 18% (9.3). All active treatments significantly reduced liver PDFF from baseline, relative changes: OM-3CA, -15%; dapagliflozin, -13%; OM-3CA + dapagliflozin, -21%. Only the combination treatment reduced liver PDFF (p = 0.046) and total liver fat volume (relative change, -24%, p = 0.037) in comparison with placebo. There was an interaction between the PNPLA3 I148M polymorphism and change in liver PDFF in the active treatment groups (p = 0.03). Dapagliflozin monotherapy, but not the combination with OM-3CA, reduced the levels of hepatocyte injury biomarkers, including alanine aminotransferase, aspartate aminotransferase, γ-glutamyl transferase (γ-GT), cytokeratin (CK) 18-M30 and CK 18-M65 and plasma fibroblast growth factor 21 (FGF21). Changes in γ-GT correlated with changes in liver PDFF (ρ = 0.53, p = 0.02). Dapagliflozin alone and in combination with OM-3CA improved glucose control and reduced body weight and abdominal fat volumes. Fatty acid oxidative stress biomarkers were not affected by treatments. There were no new or unexpected adverse events compared with previous studies with these treatments.
Conclusions/interpretation: Combined treatment with dapagliflozin and OM-3CA significantly reduced liver fat content. Dapagliflozin monotherapy reduced all measured hepatocyte injury biomarkers and FGF21, suggesting a disease-modifying effect in NAFLD.
Trial registration: ClinicalTrials.gov NCT02279407 FUNDING: The study was funded by AstraZeneca.
Keywords: Dapagliflozin; Docosahexaenoic acid; Eicosapentaenoic acid; Liver steatosis; Non-alcoholic fatty liver disease; Omega-3 fatty acids; Proton density fat fraction; Type 2 diabetes.
Conflict of interest statement
LJ is employed by Antaros Medical. MK, TM, G-BF and JO are employed by AstraZeneca. JWE, UR and LL have received consultancy fees for planning of this study. JWE has received research grants or honoraria from AstraZeneca, Bristol-Myers Squibb, Merck Sharp & Dohme, Novo Nordisk and Sanofi. All other authors declare that there is no duality of interest associated with their contribution to this manuscript.
Figures

References
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

