Cabozantinib in Patients with Advanced and Progressing Hepatocellular Carcinoma
- PMID: 29972759
- PMCID: PMC7523244
- DOI: 10.1056/NEJMoa1717002
Cabozantinib in Patients with Advanced and Progressing Hepatocellular Carcinoma
Abstract
Background: Cabozantinib inhibits tyrosine kinases, including vascular endothelial growth factor receptors 1, 2, and 3, MET, and AXL, which are implicated in the progression of hepatocellular carcinoma and the development of resistance to sorafenib, the standard initial treatment for advanced disease. This randomized, double-blind, phase 3 trial evaluated cabozantinib as compared with placebo in previously treated patients with advanced hepatocellular carcinoma.
Methods: A total of 707 patients were randomly assigned in a 2:1 ratio to receive cabozantinib (60 mg once daily) or matching placebo. Eligible patients had received previous treatment with sorafenib, had disease progression after at least one systemic treatment for hepatocellular carcinoma, and may have received up to two previous systemic regimens for advanced hepatocellular carcinoma. The primary end point was overall survival. Secondary end points were progression-free survival and the objective response rate.
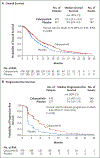
Results: At the second planned interim analysis, the trial showed significantly longer overall survival with cabozantinib than with placebo. Median overall survival was 10.2 months with cabozantinib and 8.0 months with placebo (hazard ratio for death, 0.76; 95% confidence interval [CI], 0.63 to 0.92; P=0.005). Median progression-free survival was 5.2 months with cabozantinib and 1.9 months with placebo (hazard ratio for disease progression or death, 0.44; 95% CI, 0.36 to 0.52; P<0.001), and the objective response rates were 4% and less than 1%, respectively (P=0.009). Grade 3 or 4 adverse events occurred in 68% of patients in the cabozantinib group and in 36% in the placebo group. The most common high-grade events were palmar-plantar erythrodysesthesia (17% with cabozantinib vs. 0% with placebo), hypertension (16% vs. 2%), increased aspartate aminotransferase level (12% vs. 7%), fatigue (10% vs. 4%), and diarrhea (10% vs. 2%).
Conclusions: Among patients with previously treated advanced hepatocellular carcinoma, treatment with cabozantinib resulted in longer overall survival and progression-free survival than placebo. The rate of high-grade adverse events in the cabozantinib group was approximately twice that observed in the placebo group. (Funded by Exelixis; CELESTIAL ClinicalTrials.gov number, NCT01908426 .).
Conflict of interest statement
No other potential conflict of interest relevant to this article was reported.
Figures

Comment in
-
Cabozantinib in Hepatocellular Carcinoma.N Engl J Med. 2018 Oct 4;379(14):1384. doi: 10.1056/NEJMc1810178. N Engl J Med. 2018. PMID: 30285325 No abstract available.
-
Cabozantinib and the moving field of systemic treatments in advanced hepatocellular carcinoma.Hepatobiliary Surg Nutr. 2019 Feb;8(1):53-55. doi: 10.21037/hbsn.2018.10.12. Hepatobiliary Surg Nutr. 2019. PMID: 30881965 Free PMC article. No abstract available.
-
Cabozantinib for advanced hepatocellular carcinoma.Hepatobiliary Surg Nutr. 2019 Apr;8(2):153-156. doi: 10.21037/hbsn.2018.11.22. Hepatobiliary Surg Nutr. 2019. PMID: 31098366 Free PMC article. No abstract available.
References
-
- Ferlay J, Soerjomataram I, Dikshit R, et al. Cancer incidence and mortality worldwide: sources, methods and major patterns in GLOBOCAN 2012. Int J Cancer 2015; 136(5): E359–E386. - PubMed
-
- Abou-Alfa GK, Jarnagin W, Lowery M, et al. Liver and bile duct cancer In: Niederhuber JE, Armitage JO, Doroshow JH, Kastan MB, Tepper JE, eds. Abeloff’s clinical oncology. 5th ed Philadelphia: Saunders, 2014:1373–96.
-
- National Comprehensive Cancer Network. NCCN clinical practice guidelines in oncology: hepatobiliary cancers, version 4. 2017. (https://www.nccn.org/professionals/physician_gls/pdf/hepatobiliary.pdf). - PMC - PubMed
-
- European Association for the Study of the Liver, European Organisation for Research and Treatment Of Cancer. EASL-EORTC clinical practice guidelines: management of hepatocellular carcinoma. J Hepatol 2012; 56: 908–43. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous
