Evidence for expression and functionality of FSH and LH/hCG receptors in human endometrium
- PMID: 29974367
- PMCID: PMC6133814
- DOI: 10.1007/s10815-018-1248-8
Evidence for expression and functionality of FSH and LH/hCG receptors in human endometrium
Abstract
Purpose: Follicle-stimulating hormone (FSH) and luteinizing hormone (LH) mediate intracellular functions by binding their specific protein G-coupled gonadotrophin receptor, respectively FSH receptor (FSHR) and LH/choriogonadotrophin receptor (LHCGR). Whereas the expression of FSHR and LHCGR in mammals was considered gonad-specific and cell-specific, studies identified gonadotrophin receptors in human female extragonadal reproductive tissues. This study aims to demonstrate that gonadotrophin receptors are expressed in endometrium and mediates intracellular functions.
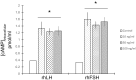
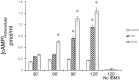
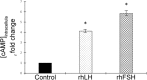
Methods: Collected endometria (n = 12) from healthy patients (mean age of 36 ± 6) were primary cultured for 24 h. The presence of gonadotrophin receptors was evaluated by RT-PCR followed by the sequencing of the resulted amplicons and by immunohistochemistry in original samples. Endometrial primary cultures were treated with increasing concentration (range 0-100 ng/ml) of either recombinant human LH (rhLH) or recombinant human FSH (rhFSH). Endometria controls had gonadotrophin replaced by the same volume of the culture medium. In gonadotrophin-treated samples, it was evaluated the intracellular cyclic adenosine monophosphate (cAMP) content by enzymatic immunoassay and the expression of steroidogenic genes by reverse transcriptase-quantitative polymerase chain reaction (RT-qPCR).
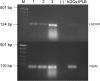
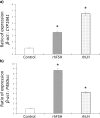
Results: The sequencing of the RT-PCR amplicons confirmed the presence of both gonadotrophin receptors and immunohistochemistry localized them on the membrane of endometrial glands cells throughout the glandular epithelium. The gonadotrophin-receptor complex was able to increase the intracellular cAMP in a dose-response and time-course manner and to induce steroidogenic genes expression.
Conclusion: This study demonstrates that both gonadotrophin receptors are expressed along the glandular epithelium of endometria and they mediate the effects of gonadotrophins on intracellular functions.
Keywords: Endometrium; FSHR; Gonadotrophin; LHCGR; Steroidogenic genes.
Conflict of interest statement
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Conflict of interest
The authors declare that they have no conflict of interest.
Figures



References
-
- Vaitukaitis JL, Ross GT, Braunstein GD, Rayford PL. Gonadotropins and their subunits: basic and clinical studies. Recent Prog Horm Res. 1976;32:289–331. - PubMed
-
- Fritz IB. Sites of action of androgens and follicle-stimulating hormone on cells of the seminiferous tubule. In: Litwack G, editor. Biochemical actions of hormones. New York: Academic; 1978. pp. 249–281.
-
- Kangasniemi M, Kaipia A, Toppari J, Perheentupa A, Huhtaniemi I, Parvinen M. Cellular regulation of follicle-stimulating hormone (FSH) binding in rat seminiferous tubules. J Androl. 1990;11(4):336–343. - PubMed
-
- Dankbar B, Brinkworth MH, Schlatt S, Weinbauer GF, Nieschlag E, Gromoll J. Ubiquitous expression of the androgen receptor and testis-specific expression of the FSH receptor in the cynomolgus monkey (Macaca fascicularis) revealed by a ribonuclease protection assay. J Steroid Biochem Mol Biol. 1995;55(1):35–41. doi: 10.1016/0960-0760(95)00148-S. - DOI - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

