Localized Induction of Wild-Type and Mutant Alpha-Synuclein Aggregation Reveals Propagation along Neuroanatomical Tracts
- PMID: 29976670
- PMCID: PMC6146706
- DOI: 10.1128/JVI.00586-18
Localized Induction of Wild-Type and Mutant Alpha-Synuclein Aggregation Reveals Propagation along Neuroanatomical Tracts
Abstract
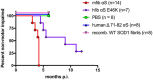
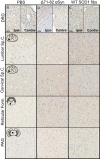
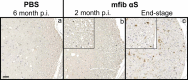
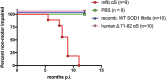
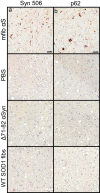
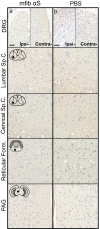
Misfolded alpha-synuclein (αS) may exhibit a number of characteristics similar to those of the prion protein, including the apparent ability to spread along neuroanatomical connections. The demonstration for this mechanism of spread is largely based on the intracerebral injections of preaggregated αS seeds in mice, in which it cannot be excluded that diffuse, surgical perturbations and hematogenous spread also contribute to the propagation of pathology. For this reason, we have utilized the sciatic nerve as a route of injection to force the inoculum into the lumbar spinal cord and induce a localized site for the onset of αS inclusion pathology. Our results demonstrate that mouse αS fibrils (fibs) injected unilaterally in the sciatic nerve are efficient in inducing pathology and the onset of paralytic symptoms in both the M83 and M20 lines of αS transgenic mice. In addition, a spatiotemporal study of these injections revealed a predictable spread of pathology to brain regions whose axons synapse directly on ventral motor neurons in the spinal cord, strongly supporting axonal transport as a mechanism of spread of the αS inducing, or seeding, factor. We also revealed a relatively decreased efficiency for human αS fibs containing the E46K mutation to induce disease via this injection paradigm, supportive of recent studies demonstrating a diminished ability of this mutant αS to undergo aggregate induction. These results further demonstrate prion-like properties for αS by the ability for a progression and spread of αS inclusion pathology along neuroanatomical connections.IMPORTANCE The accumulation of alpha-synuclein (αS) inclusions is a hallmark feature of Parkinson's disease (PD) and PD-related diseases. Recently, a number of studies have demonstrated similarities between the prion protein and αS, including its ability to spread along neuroanatomical tracts throughout the central nervous system (CNS). However, there are caveats in each of these studies in which the injection routes used had the potential to result in a widespread dissemination of the αS-containing inocula, making it difficult to precisely define the mechanisms of spread. In this study, we assessed the spread of pathology following a localized induction of αS inclusions in the lumbar spinal cord following a unilateral injection in the sciatic nerve. Using this paradigm, we demonstrated the ability for αS inclusion spread and/or induction along neuroanatomical tracts within the CNS of two αS-overexpressing mouse models.
Keywords: alpha-synuclein; axonal transport; prion; prions; propagation; sciatic nerve; α-synuclein.
Copyright © 2018 American Society for Microbiology.
Figures





Similar articles
-
Robust Central Nervous System Pathology in Transgenic Mice following Peripheral Injection of α-Synuclein Fibrils.J Virol. 2017 Jan 3;91(2):e02095-16. doi: 10.1128/JVI.02095-16. Print 2017 Jan 15. J Virol. 2017. PMID: 27852849 Free PMC article.
-
Comparison of the in vivo induction and transmission of α-synuclein pathology by mutant α-synuclein fibril seeds in transgenic mice.Hum Mol Genet. 2017 Dec 15;26(24):4906-4915. doi: 10.1093/hmg/ddx371. Hum Mol Genet. 2017. PMID: 29036344 Free PMC article.
-
Comparative analyses of the in vivo induction and transmission of α-synuclein pathology in transgenic mice by MSA brain lysate and recombinant α-synuclein fibrils.Acta Neuropathol Commun. 2019 May 20;7(1):80. doi: 10.1186/s40478-019-0733-3. Acta Neuropathol Commun. 2019. PMID: 31109378 Free PMC article.
-
[Animal models of synucleinopathies: prion-like propagation of alpha-synuclein in wild-type animals].Nihon Yakurigaku Zasshi. 2019;154(6):301-305. doi: 10.1254/fpj.154.301. Nihon Yakurigaku Zasshi. 2019. PMID: 31787680 Review. Japanese.
-
Propagation of alpha-synuclein pathology: hypotheses, discoveries, and yet unresolved questions from experimental and human brain studies.Acta Neuropathol. 2016 Jan;131(1):49-73. doi: 10.1007/s00401-015-1485-1. Epub 2015 Oct 7. Acta Neuropathol. 2016. PMID: 26446103 Free PMC article. Review.
Cited by
-
Prodromal neuroinvasion of pathological α-synuclein in brainstem reticular nuclei and white matter lesions in a model of α-synucleinopathy.Brain Commun. 2021 May 14;3(2):fcab104. doi: 10.1093/braincomms/fcab104. eCollection 2021. Brain Commun. 2021. PMID: 34136810 Free PMC article.
-
MiR-30c-5p/ATG5 Axis Regulates the Progression of Parkinson's Disease.Front Cell Neurosci. 2021 May 25;15:644507. doi: 10.3389/fncel.2021.644507. eCollection 2021. Front Cell Neurosci. 2021. PMID: 34113238 Free PMC article.
-
Carboxy-terminal truncation and phosphorylation of α-synuclein elongates survival in a prion-like seeding mouse model of synucleinopathy.Neurosci Lett. 2020 Jul 27;732:135017. doi: 10.1016/j.neulet.2020.135017. Epub 2020 May 1. Neurosci Lett. 2020. PMID: 32371157 Free PMC article.
-
Trans-synaptic spreading of alpha-synuclein pathology through sensory afferents leads to sensory nerve degeneration and neuropathic pain.Acta Neuropathol Commun. 2021 Feb 25;9(1):31. doi: 10.1186/s40478-021-01131-8. Acta Neuropathol Commun. 2021. PMID: 33632316 Free PMC article.
-
Bradykinesia and postural instability in a model of prodromal synucleinopathy with α-synuclein aggregation initiated in the gigantocellular nuclei.Acta Neuropathol Commun. 2025 Feb 17;13(1):32. doi: 10.1186/s40478-025-01948-7. Acta Neuropathol Commun. 2025. PMID: 39962601 Free PMC article.
References
-
- Goedert M. 1997. Familial Parkinson's disease. The awakening of alpha-synuclein. Nature 388:232–233. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

