CD32+ and PD-1+ Lymph Node CD4 T Cells Support Persistent HIV-1 Transcription in Treated Aviremic Individuals
- PMID: 29976671
- PMCID: PMC6158413
- DOI: 10.1128/JVI.00901-18
CD32+ and PD-1+ Lymph Node CD4 T Cells Support Persistent HIV-1 Transcription in Treated Aviremic Individuals
Abstract
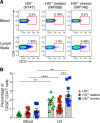
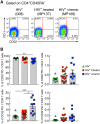
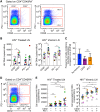
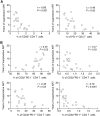
A recent study conducted in blood has proposed CD32 as the marker identifying the "elusive" HIV reservoir. We have investigated the distribution of CD32+ CD4 T cells in blood and lymph nodes (LNs) of HIV-1-uninfected subjects and viremic untreated and long-term-treated HIV-1-infected individuals and their relationship with PD-1+ CD4 T cells. The frequency of CD32+ CD4 T cells was increased in viremic compared to treated individuals in LNs, and a large proportion (up to 50%) of CD32+ cells coexpressed PD-1 and were enriched within T follicular helper (Tfh) cells. We next investigated the role of LN CD32+ CD4 T cells in the HIV reservoir. Total HIV DNA was enriched in CD32+ and PD-1+ CD4 T cells compared to CD32- and PD-1- cells in both viremic and treated individuals, but there was no difference between CD32+ and PD-1+ cells. There was no enrichment of latently infected cells with inducible HIV-1 in CD32+ versus PD-1+ cells in antiretroviral therapy (ART)-treated individuals. HIV-1 transcription was then analyzed in LN memory CD4 T cell populations sorted on the basis of CD32 and PD-1 expression. CD32+ PD-1+ CD4 T cells were significantly enriched in cell-associated HIV RNA compared to CD32- PD-1- (averages of 5.2-fold in treated individuals and 86.6-fold in viremics), CD32+ PD-1- (2.2-fold in treated individuals and 4.3-fold in viremics), and CD32- PD-1+ (2.2-fold in ART-treated individuals and 4.6-fold in viremics) cell populations. Similar levels of HIV-1 transcription were found in CD32+ PD-1- and CD32- PD-1+ CD4 T cells. Interestingly, the proportion of CD32+ and PD-1+ CD4 T cells negatively correlated with CD4 T cell counts and length of therapy. Therefore, the expression of CD32 identifies, independently of PD-1, a CD4 T cell population with persistent HIV-1 transcription and coexpression of CD32 and PD-1, the CD4 T cell population with the highest levels of HIV-1 transcription in both viremic and treated individuals.IMPORTANCE The existence of long-lived latently infected resting memory CD4 T cells represents a major obstacle to the eradication of HIV infection. Identifying cell markers defining latently infected cells containing replication-competent virus is important in order to determine the mechanisms of HIV persistence and to develop novel therapeutic strategies to cure HIV infection. We provide evidence that PD-1 and CD32 may have a complementary role in better defining CD4 T cell populations infected with HIV-1. Furthermore, CD4 T cells coexpressing CD32 and PD-1 identify a CD4 T cell population with high levels of persistent HIV-1 transcription.
Keywords: CD32; PD-1; Tfh cells; human immunodeficiency virus; lymph node.
Copyright © 2018 Noto et al.
Figures



References
-
- Chun TW, Carruth L, Finzi D, Shen X, DiGiuseppe JA, Taylor H, Hermankova M, Chadwick K, Margolick J, Quinn TC, Kuo YH, Brookmeyer R, Zeiger MA, Barditch-Crovo P, Siliciano RF. 1997. Quantification of latent tissue reservoirs and total body viral load in HIV-1 infection. Nature 387:183–188. doi: 10.1038/387183a0. - DOI - PubMed
-
- Finzi D, Hermankova M, Pierson T, Carruth LM, Buck C, Chaisson RE, Quinn TC, Chadwick K, Margolick J, Brookmeyer R, Gallant J, Markowitz M, Ho DD, Richman DD, Siliciano RF. 1997. Identification of a reservoir for HIV-1 in patients on highly active antiretroviral therapy. Science 278:1295–1300. doi: 10.1126/science.278.5341.1295. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

