11C-DPA-713 Versus 18F-GE-180: A Preclinical Comparison of Translocator Protein 18 kDa PET Tracers to Visualize Acute and Chronic Neuroinflammation in a Mouse Model of Ischemic Stroke
- PMID: 29976695
- PMCID: PMC6354224
- DOI: 10.2967/jnumed.118.209155
11C-DPA-713 Versus 18F-GE-180: A Preclinical Comparison of Translocator Protein 18 kDa PET Tracers to Visualize Acute and Chronic Neuroinflammation in a Mouse Model of Ischemic Stroke
Abstract
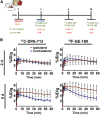
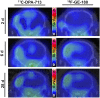
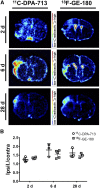
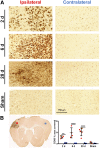
Neuroinflammation plays a key role in neuronal injury after ischemic stroke. PET imaging of translocator protein 18 kDa (TSPO) permits longitudinal, noninvasive visualization of neuroinflammation in both preclinical and clinical settings. Many TSPO tracers have been developed, however, it is unclear which tracer is the most sensitive and accurate for monitoring the in vivo spatiotemporal dynamics of neuroinflammation across applications. Hence, there is a need for head-to-head comparisons of promising TSPO PET tracers across different disease states. Accordingly, the aim of this study was to directly compare 2 promising second-generation TSPO tracers, 11C-DPA-713 and 18F-GE-180, for the first time at acute and chronic time points after ischemic stroke. Methods: After distal middle cerebral artery occlusion or sham surgery, mice underwent consecutive PET/CT imaging with 11C-DPA-713 and 18F-GE-180 at 2, 6, and 28 d after stroke. T2-weighted MR images were acquired to enable delineation of ipsilateral (infarct) and contralateral brain regions of interest (ROIs). PET/CT images were analyzed by calculating percentage injected dose per gram in MR-guided ROIs. SUV ratios were determined using the contralateral thalamus (SUVTh) as a pseudoreference region. Ex vivo autoradiography and immunohistochemistry were performed to verify in vivo findings. Results: Significantly increased tracer uptake was observed in the ipsilateral compared with contralateral ROI (SUVTh, 50-60 min summed data) at acute and chronic time points using 11C-DPA-713 and 18F-GE-180. Ex vivo autoradiography confirmed in vivo findings demonstrating increased TSPO tracer uptake in infarcted versus contralateral brain tissue. Importantly, a significant correlation was identified between microglial/macrophage activation (cluster of differentiation 68 immunostaining) and 11C-DPA-713- PET signal, which was not evident with 18F-GE-180. No significant correlations were observed between TSPO PET and activated astrocytes (glial fibrillary acidic protein immunostaining). Conclusion:11C-DPA-713 and 18F-GE-180 PET enable detection of neuroinflammation at acute and chronic time points after cerebral ischemia in mice. 11C-DPA-713 PET reflects the extent of microglial activation in infarcted distal middle cerebral artery occlusion mouse brain tissue more accurately than 18F-GE-180 and appears to be slightly more sensitive. These results highlight the potential of 11C-DPA-713 for tracking microglial activation in vivo after stroke and warrant further investigation in both preclinical and clinical settings.
Keywords: PET; TSPO; ischemic stroke; neuroinflammation.
© 2019 by the Society of Nuclear Medicine and Molecular Imaging.
Figures


Similar articles
-
Comparative Evaluation of Three TSPO PET Radiotracers in a LPS-Induced Model of Mild Neuroinflammation in Rats.Mol Imaging Biol. 2017 Feb;19(1):77-89. doi: 10.1007/s11307-016-0984-3. Mol Imaging Biol. 2017. PMID: 27481358 Free PMC article.
-
Development and evaluation of [11C]DPA-813 and [18F]DPA-814: novel TSPO PET tracers insensitive to human single nucleotide polymorphism rs6971.Eur J Nucl Med Mol Imaging. 2025 Jun;52(7):2658-2670. doi: 10.1007/s00259-025-07109-1. Epub 2025 Feb 5. Eur J Nucl Med Mol Imaging. 2025. PMID: 39907797 Free PMC article.
-
Detection of microglial activation in an acute model of neuroinflammation using PET and radiotracers 11C-(R)-PK11195 and 18F-GE-180.J Nucl Med. 2014 Mar;55(3):466-72. doi: 10.2967/jnumed.113.125625. Epub 2014 Feb 10. J Nucl Med. 2014. PMID: 24516258
-
The potential of carbon-11 and fluorine-18 chemistry: illustration through the development of positron emission tomography radioligands targeting the translocator protein 18 kDa.J Labelled Comp Radiopharm. 2013 Mar-Apr;56(3-4):96-104. doi: 10.1002/jlcr.2992. J Labelled Comp Radiopharm. 2013. PMID: 24285315 Review.
-
TSPO imaging in animal models of brain diseases.Eur J Nucl Med Mol Imaging. 2021 Dec;49(1):77-109. doi: 10.1007/s00259-021-05379-z. Epub 2021 Jul 10. Eur J Nucl Med Mol Imaging. 2021. PMID: 34245328 Free PMC article. Review.
Cited by
-
Direct Comparison of [18F]F-DPA with [18F]DPA-714 and [11C]PBR28 for Neuroinflammation Imaging in the same Alzheimer's Disease Model Mice and Healthy Controls.Mol Imaging Biol. 2022 Feb;24(1):157-166. doi: 10.1007/s11307-021-01646-5. Epub 2021 Sep 20. Mol Imaging Biol. 2022. PMID: 34542805 Free PMC article.
-
TSPO PET detects acute neuroinflammation but not diffuse chronically activated MHCII microglia in the rat.EJNMMI Res. 2020 Sep 29;10(1):113. doi: 10.1186/s13550-020-00699-x. EJNMMI Res. 2020. PMID: 32990808 Free PMC article.
-
Letter to the Editor re: Confirmation of Specific Binding of the 18-kDa Translocator Protein (TSPO) Radioligand [18F]GE-180: a Blocking Study Using XBD173 in Multiple Sclerosis Normal Appearing White and Grey Matter.Mol Imaging Biol. 2020 Feb;22(1):10-12. doi: 10.1007/s11307-019-01433-3. Mol Imaging Biol. 2020. PMID: 31641965 No abstract available.
-
Translatome analysis reveals microglia and astrocytes to be distinct regulators of inflammation in the hyperacute and acute phases after stroke.Glia. 2023 Aug;71(8):1960-1984. doi: 10.1002/glia.24377. Epub 2023 Apr 17. Glia. 2023. PMID: 37067534 Free PMC article.
-
Essential Principles and Recent Progress in the Development of TSPO PET Ligands for Neuroinflammation Imaging.Curr Med Chem. 2022 Aug 6;29(28):4862-4890. doi: 10.2174/0929867329666220329204054. Curr Med Chem. 2022. PMID: 35352645 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
