Improving catalytic activity of the Baeyer-Villiger monooxygenase-based Escherichia coli biocatalysts for the overproduction of (Z)-11-(heptanoyloxy)undec-9-enoic acid from ricinoleic acid
- PMID: 29980730
- PMCID: PMC6035261
- DOI: 10.1038/s41598-018-28575-8
Improving catalytic activity of the Baeyer-Villiger monooxygenase-based Escherichia coli biocatalysts for the overproduction of (Z)-11-(heptanoyloxy)undec-9-enoic acid from ricinoleic acid
Abstract
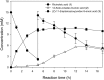
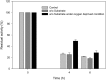
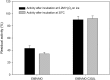
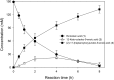
Baeyer-Villiger monooxygenases (BVMOs) can be used for the biosynthesis of lactones and esters from ketones. However, the BVMO-based biocatalysts are not so stable under process conditions. Thereby, this study focused on enhancing stability of the BVMO-based biocatalysts. The biotransformation of ricinoleic acid into (Z)-11-(heptanoyloxy)undec-9-enoic acid by the recombinant Escherichia coli expressing the BVMO from Pseudomonas putida and an alcohol dehydrogenase from Micrococcus luteus was used as a model system. After thorough investigation of the key factors to influence stability of the BVMO, Cys302 was identified as an engineering target. The substitution of Cys302 to Leu enabled the engineered enzyme (i.e., E6BVMOC302L) to become more stable toward oxidative and thermal stresses. The catalytic activity of E6BVMOC302L-based E. coli biocatalysts was also greater than the E6BVMO-based biocatalysts. Another factor to influence biocatalytic performance of the BVMO-based whole-cell biocatalysts was availability of carbon and energy source during biotransformations. Glucose feeding into the reaction medium led to a marked increase of final product concentrations. Overall, the bioprocess engineering to improve metabolic stability of host cells in addition to the BVMO engineering allowed us to produce (Z)-11-(heptanoyloxy)undec-9-enoic acid to a concentration of 132 mM (41 g/L) from 150 mM ricinoleic acid within 8 h.
Conflict of interest statement
The authors declare no competing interests.
Figures


Similar articles
-
Engineering of Baeyer-Villiger monooxygenase-based Escherichia coli biocatalyst for large scale biotransformation of ricinoleic acid into (Z)-11-(heptanoyloxy)undec-9-enoic acid.Sci Rep. 2016 Jun 17;6:28223. doi: 10.1038/srep28223. Sci Rep. 2016. PMID: 27311560 Free PMC article.
-
Production of (Z)-11-(heptanoyloxy)undec-9-enoic acid from ricinoleic acid by utilizing crude glycerol as sole carbon source in engineered Escherichia coli expressing BVMO-ADH-FadL.Enzyme Microb Technol. 2018 Dec;119:45-51. doi: 10.1016/j.enzmictec.2018.09.001. Epub 2018 Sep 5. Enzyme Microb Technol. 2018. PMID: 30243386
-
Continuous supply of glucose and glycerol enhances biotransformation of ricinoleic acid to (E)-11-(heptanoyloxy) undec-9-enoic acid in recombinant Escherichia coli.J Biotechnol. 2017 Jul 10;253:34-39. doi: 10.1016/j.jbiotec.2017.05.014. Epub 2017 May 20. J Biotechnol. 2017. PMID: 28536060
-
Recent developments in the application of Baeyer-Villiger monooxygenases as biocatalysts.Chembiochem. 2010 Nov 2;11(16):2208-31. doi: 10.1002/cbic.201000395. Chembiochem. 2010. PMID: 20936617 Review.
-
Baeyer-Villiger monooxygenases: recent advances and future challenges.Curr Opin Chem Biol. 2010 Apr;14(2):138-44. doi: 10.1016/j.cbpa.2009.11.017. Epub 2009 Dec 16. Curr Opin Chem Biol. 2010. PMID: 20015679 Review.
Cited by
-
Unleashing the Synthetic Power of Plant Oxygenases: From Mechanism to Application.Plant Physiol. 2019 Mar;179(3):813-829. doi: 10.1104/pp.18.01223. Epub 2019 Jan 22. Plant Physiol. 2019. PMID: 30670605 Free PMC article. Review.
-
Exploitation of Hetero- and Phototrophic Metabolic Modules for Redox-Intensive Whole-Cell Biocatalysis.Front Bioeng Biotechnol. 2022 Apr 13;10:855715. doi: 10.3389/fbioe.2022.855715. eCollection 2022. Front Bioeng Biotechnol. 2022. PMID: 35497353 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

