Small Molecules Identified from a Quantitative Drug Combinational Screen Resensitize Cisplatin's Response in Drug-Resistant Ovarian Cancer Cells
- PMID: 29982103
- PMCID: PMC6034569
- DOI: 10.1016/j.tranon.2018.06.002
Small Molecules Identified from a Quantitative Drug Combinational Screen Resensitize Cisplatin's Response in Drug-Resistant Ovarian Cancer Cells
Abstract
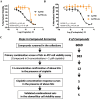
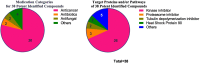
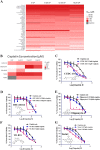
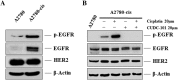
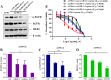
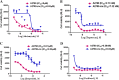
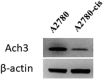
Drug resistance to chemotherapy occurs in many ovarian cancer patients resulting in failure of treatment. Exploration of drug resistance mechanisms and identification of new therapeutics that overcome the drug resistance can improve patient prognosis. Following a quantitative combination screen of 6060 approved drugs and bioactive compounds in a cisplatin-resistant A2780-cis ovarian cancer cell line, 38 active compounds with IC50s under 1 μM suppressed the growth of cisplatin-resistant ovarian cancer cells. Among these confirmed compounds, CUDC-101, OSU-03012, oligomycin A, VE-821, or Torin2 in a combination with cisplatin restored cisplatin's apoptotic response in the A2780-cis cells, while SR-3306, GSK-923295, SNX-5422, AT-13387, and PF-05212384 directly suppressed the growth of A2780-cis cells. One of the mechanisms for overcoming cisplatin resistance in these cells is mediated by the inhibition of epidermal growth factor receptor (EGFR), though not all the EGFR inhibitors are equally active. The increased levels of total EGFR and phosphorylated-EGFR (p-EGFR) in the A2780-cis cells were reduced after the combined treatment of cisplatin with EGFR inhibitors. In addition, a knockdown of EGFR mRNA reduced cisplatin resistance in the A2780-cis cells. Therefore, the top active compounds identified in this work can be studied further as potential treatments for cisplatin-resistant ovarian cancer. The quantitative combinational screening approach is a useful method for identifying effective compounds and drug combinations against drug-resistant cancer cells.
Copyright © 2018 The Authors. Published by Elsevier Inc. All rights reserved.
Figures



Similar articles
-
The role of topotecan for extending the platinum-free interval in recurrent ovarian cancer: an in vitro model.Gynecol Oncol. 2004 Jul;94(1):67-73. doi: 10.1016/j.ygyno.2004.03.047. Gynecol Oncol. 2004. PMID: 15262121
-
New and Old Genes Associated with Primary and Established Responses to Cisplatin and Topotecan Treatment in Ovarian Cancer Cell Lines.Molecules. 2017 Oct 13;22(10):1717. doi: 10.3390/molecules22101717. Molecules. 2017. PMID: 29027969 Free PMC article.
-
In vitro studies on the mechanisms of oxaliplatin resistance.Cancer Chemother Pharmacol. 2001 Nov;48(5):398-406. doi: 10.1007/s002800100363. Cancer Chemother Pharmacol. 2001. PMID: 11761458
-
The integration of paclitaxel and new platinum compounds in the treatment of advanced ovarian cancer.Int J Gynecol Cancer. 2001;11 Suppl 1:21-30. doi: 10.1046/j.1525-1438.2001.11(suppl.1)sup1021.x. Int J Gynecol Cancer. 2001. PMID: 11488999 Review.
-
Cisplatin for cancer therapy and overcoming chemoresistance.Heliyon. 2022 Sep 14;8(9):e10608. doi: 10.1016/j.heliyon.2022.e10608. eCollection 2022 Sep. Heliyon. 2022. PMID: 36158077 Free PMC article. Review.
Cited by
-
Therapeutic strategies to induce ERα in luminal breast cancer to enhance tamoxifen efficacy.Endocr Relat Cancer. 2019 Aug;26(8):689-698. doi: 10.1530/ERC-19-0042. Endocr Relat Cancer. 2019. PMID: 31167163 Free PMC article.
-
Celecoxib Analogues for Cancer Treatment: An Update on OSU-03012 and 2,5-Dimethyl-Celecoxib.Biomolecules. 2021 Jul 16;11(7):1049. doi: 10.3390/biom11071049. Biomolecules. 2021. PMID: 34356673 Free PMC article. Review.
-
Effectiveness and safety of mono-anlotinib mono therapy or in combination with chemotherapy in platinum-resistant recurrent ovarian cancer: a single-center retrospective study.Am J Transl Res. 2023 Mar 15;15(3):1973-1981. eCollection 2023. Am J Transl Res. 2023. PMID: 37056822 Free PMC article.
-
CENPE Inhibition Leads to Mitotic Catastrophe and DNA Damage in Medulloblastoma Cells.Cancers (Basel). 2021 Mar 1;13(5):1028. doi: 10.3390/cancers13051028. Cancers (Basel). 2021. PMID: 33804489 Free PMC article.
-
BRAF Inhibitor Resistance Confers Increased Sensitivity to Mitotic Inhibitors.Front Oncol. 2022 Apr 4;12:766794. doi: 10.3389/fonc.2022.766794. eCollection 2022. Front Oncol. 2022. PMID: 35444937 Free PMC article.
References
-
- Raja FA, Chopra N, Ledermann JA. Optimal first-line treatment in ovarian cancer. Ann Oncol. 2012;23(Suppl. 10):x118–x127. - PubMed
-
- Siegel R, Naishadham D, Jemal A. Cancer statistics, 2012. CA Cancer J Clin. 2012;62:10–29. - PubMed
-
- du Bois A, Weber B, Rochon J, Meier W, Goupil A, Olbricht S, Barats JC, Kuhn W, Orfeuvre H, Wagner U. Addition of epirubicin as a third drug to carboplatin-paclitaxel in first-line treatment of advanced ovarian cancer: a prospectively randomized gynecologic cancer intergroup trial by the Arbeitsgemeinschaft Gynaekologische Onkologie Ovarian Cancer Study Group and the Groupe d'Investigateurs Nationaux pour l'Etude des Cancers Ovariens. J Clin Oncol. 2006;24:1127–1135. - PubMed
-
- Bookman MA, Brady MF, McGuire WP, Harper PG, Alberts DS, Friedlander M, Colombo N, Fowler JM, Argenta PA, De Geest K. Evaluation of new platinum-based treatment regimens in advanced-stage ovarian cancer: a Phase III Trial of the Gynecologic Cancer Intergroup. J Clin Oncol. 2009;27:1419–1425. - PMC - PubMed
-
- Galluzzi L, Senovilla L, Vitale I, Michels J, Martins I, Kepp O, Castedo M, Kroemer G. Molecular mechanisms of cisplatin resistance. Oncogene. 2012;31:1869–1883. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

