Intra-arterial Versus Intravenous Adoptive Cell Therapy in a Mouse Tumor Model
- PMID: 29985207
- PMCID: PMC6092084
- DOI: 10.1097/CJI.0000000000000235
Intra-arterial Versus Intravenous Adoptive Cell Therapy in a Mouse Tumor Model
Abstract
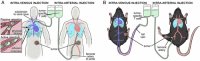
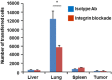
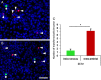
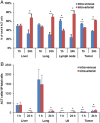
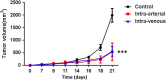
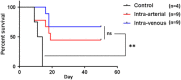
Adoptive cell transfer therapy for cancer has existed for decades and is experiencing a resurgence in popularity that has been facilitated by improved methods of production, techniques for genetic modification, and host preconditioning. The trafficking of adoptively transferred lymphocytes and infiltration into the tumor microenvironment is sine qua non for successful tumor eradication; however, the paradox of extremely poor trafficking of lymphocytes into the tumor microenvironment raises the issue of how best to deliver these cells to optimize entry into tumor tissue. We examined the route of administration as a potential modifier of both trafficking and antitumor efficacy. Femoral artery cannulation and tail vein injection for the intra-arterial (IA) and IV delivery, respectively, were utilized in the B16-OVA/OT-I mouse model system. Both IV and IA infusions showed decreased tumor growth and prolonged survival. However, although significantly increased T-cell tumor infiltration was observed in IA mice, tumor growth and survival were not improved as compared with IV mice. These studies suggest that IA administration produces increased early lymphocyte trafficking, but a discernable survival benefit was not seen in the murine model examined.
Figures

References
-
- Rosenberg SA, Packard BS, Aebersold PM, et al. Use of tumor-infiltrating lymphocytes and interleukin-2 in the immunotherapy of patients with metastatic melanoma. A preliminary report. N Engl J Med. 1988;319:1676–1680. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

