Recent developments in synthetic biology and metabolic engineering in microalgae towards biofuel production
- PMID: 29988523
- PMCID: PMC6026345
- DOI: 10.1186/s13068-018-1181-1
Recent developments in synthetic biology and metabolic engineering in microalgae towards biofuel production
Abstract
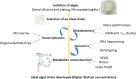
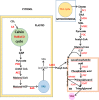
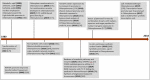
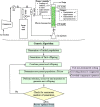
In the wake of the uprising global energy crisis, microalgae have emerged as an alternate feedstock for biofuel production. In addition, microalgae bear immense potential as bio-cell factories in terms of producing key chemicals, recombinant proteins, enzymes, lipid, hydrogen and alcohol. Abstraction of such high-value products (algal biorefinery approach) facilitates to make microalgae-based renewable energy an economically viable option. Synthetic biology is an emerging field that harmoniously blends science and engineering to help design and construct novel biological systems, with an aim to achieve rationally formulated objectives. However, resources and tools used for such nuclear manipulation, construction of synthetic gene network and genome-scale reconstruction of microalgae are limited. Herein, we present recent developments in the upcoming field of microalgae employed as a model system for synthetic biology applications and highlight the importance of genome-scale reconstruction models and kinetic models, to maximize the metabolic output by understanding the intricacies of algal growth. This review also examines the role played by microalgae as biorefineries, microalgal culture conditions and various operating parameters that need to be optimized to yield biofuel that can be economically competitive with fossil fuels.
Keywords: Biofuel; Biorefinery; Genome-scale reconstruction; Microalgae; Optimization models; Synthetic biology.
Figures
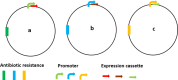
References
-
- Shuba ES, Kifle D. Microalgae to biofuels: ‘promising’alternative and renewable energy, review. Renew Sustain Energy Rev. 2018;81:743–755. doi: 10.1016/j.rser.2017.08.042. - DOI
-
- Acheampong M, Ertem FC, Kappler B, Neubauer P. In pursuit of sustainable development goal (SDG) number 7: will biofuels be reliable? Renew Sustain Energy Rev. 2017;75:927–937. doi: 10.1016/j.rser.2016.11.074. - DOI
-
- Gaurav N, Sivasankari S, Kiran GS, Ninawe A, Selvin J. Utilization of bioresources for sustainable biofuels: a review. Renew Sustain Energy Rev. 2017;73:205–214. doi: 10.1016/j.rser.2017.01.070. - DOI
-
- Ahmad AL, Yasin NHM, Derek CJC, Lim JK. Microalgae as a sustainable energy source for biodiesel production: a review. Renew Sustain Energy Rev. 2011;15:584–593. doi: 10.1016/j.rser.2010.09.018. - DOI
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources

