Real-time optical manipulation of cardiac conduction in intact hearts
- PMID: 29989169
- PMCID: PMC6117584
- DOI: 10.1113/JP276283
Real-time optical manipulation of cardiac conduction in intact hearts
Abstract
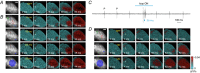
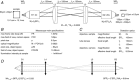
Key points: Although optogenetics has clearly demonstrated the feasibility of cardiac manipulation, current optical stimulation strategies lack the capability to react acutely to ongoing cardiac wave dynamics. Here, we developed an all-optical platform to monitor and control electrical activity in real-time. The methodology was applied to restore normal electrical activity after atrioventricular block and to manipulate the intraventricular propagation of the electrical wavefront. The closed-loop approach was also applied to simulate a re-entrant circuit across the ventricle. The development of this innovative optical methodology provides the first proof-of-concept that a real-time all-optical stimulation can control cardiac rhythm in normal and abnormal conditions.
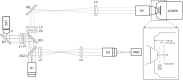
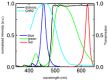
Abstract: Optogenetics has provided new insights in cardiovascular research, leading to new methods for cardiac pacing, resynchronization therapy and cardioversion. Although these interventions have clearly demonstrated the feasibility of cardiac manipulation, current optical stimulation strategies do not take into account cardiac wave dynamics in real time. Here, we developed an all-optical platform complemented by integrated, newly developed software to monitor and control electrical activity in intact mouse hearts. The system combined a wide-field mesoscope with a digital projector for optogenetic activation. Cardiac functionality could be manipulated either in free-run mode with submillisecond temporal resolution or in a closed-loop fashion: a tailored hardware and software platform allowed real-time intervention capable of reacting within 2 ms. The methodology was applied to restore normal electrical activity after atrioventricular block, by triggering the ventricle in response to optically mapped atrial activity with appropriate timing. Real-time intraventricular manipulation of the propagating electrical wavefront was also demonstrated, opening the prospect for real-time resynchronization therapy and cardiac defibrillation. Furthermore, the closed-loop approach was applied to simulate a re-entrant circuit across the ventricle demonstrating the capability of our system to manipulate heart conduction with high versatility even in arrhythmogenic conditions. The development of this innovative optical methodology provides the first proof-of-concept that a real-time optically based stimulation can control cardiac rhythm in normal and abnormal conditions, promising a new approach for the investigation of the (patho)physiology of the heart.
Keywords: Cardiac electrophysiology; Digital Micromirror Device; Optical mapping; Optogenetics.
© 2018 The Authors The Journal of Physiology published by John Wiley & Sons Ltd on behalf of The Physiological Society.
Figures







References
-
- Altomare C, Bartolucci C, Sala L, Bernardi J, Mostacciuolo G, Rocchetti M, Severi S & Zaza A (2015). IKr Impact on Repolarization and Its Variability Assessed by Dynamic Clamp. Circ Arrhythm Electrophysiol 8, 1265–1275. - PubMed
-
- Arrenberg AB, Stainier DY, Baier H & Huisken J (2010). Optogenetic control of cardiac function. Science 330, 971–974. - PubMed
-
- Baruscotti M, Bucchi A, Viscomi C, Mandelli G, Consalez G, Gnecchi‐Rusconi T, Montano N, Casali KR, Micheloni S, Barbuti A & DiFrancesco D (2011). Deep bradycardia and heart block caused by inducible cardiac‐specific knockout of the pacemaker channel gene Hcn4. Proc Natl Acad Sci U S A 108, 1705–1710. - PMC - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

