Maternal overnutrition by hypercaloric diets programs hypothalamic mitochondrial fusion and metabolic dysfunction in rat male offspring
- PMID: 29991958
- PMCID: PMC5987395
- DOI: 10.1186/s12986-018-0279-6
Maternal overnutrition by hypercaloric diets programs hypothalamic mitochondrial fusion and metabolic dysfunction in rat male offspring
Abstract
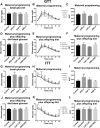
Background: Maternal overnutrition including pre-pregnancy, pregnancy and lactation promotes a lipotoxic insult leading to metabolic dysfunction in offspring. Diet-induced obesity models (DIO) show that changes in hypothalamic mitochondria fusion and fission dynamics modulate metabolic dysfunction. Using three selective diet formula including a High fat diet (HFD), Cafeteria (CAF) and High Sugar Diet (HSD), we hypothesized that maternal diets exposure program leads to selective changes in hypothalamic mitochondria fusion and fission dynamics in male offspring leading to metabolic dysfunction which is exacerbated by a second exposure after weaning.
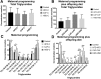
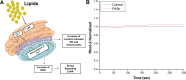
Methods: We exposed female Wistar rats to nutritional programming including Chow, HFD, CAF, or HSD for 9 weeks (pre-mating, mating, pregnancy and lactation) or to the same diets to offspring after weaning. We determined body weight, food intake and metabolic parameters in the offspring from 21 to 60 days old. Hypothalamus was dissected at 60 days old to determine mitochondria-ER interaction markers by mRNA expression and western blot and morphology by transmission electron microscopy (TEM). Mitochondrial-ER function was analyzed by confocal microscopy using hypothalamic cell line mHypoA-CLU192.
Results: Maternal programming by HFD and CAF leads to failure in glucose, leptin and insulin sensitivity and fat accumulation. Additionally, HFD and CAF programming promote mitochondrial fusion by increasing the expression of MFN2 and decreasing DRP1, respectively. Further, TEM analysis confirms that CAF exposure after programing leads to an increase in mitochondria fusion and enhanced mitochondrial-ER interaction, which partially correlates with metabolic dysfunction and fat accumulation in the HFD and CAF groups. Finally, we identified that lipotoxic palmitic acid stimulus in hypothalamic cells increases Ca2+ overload into mitochondria matrix leading to mitochondrial dysfunction.
Conclusions: We concluded that maternal programming by HFD induces hypothalamic mitochondria fusion, metabolic dysfunction and fat accumulation in male offspring, which is exacerbated by HFD or CAF exposure after weaning, potentially due to mitochondria calcium overflux.
Keywords: Diet induced obesity (DIO); Fission; Fusion; Hypothalamus; Maternal overnutrition; Mitochondria; Mitochondria dynamics.
Conflict of interest statement
The procedures for the care and use of experimental animals followed the protocols and regulations set forth by Animal Care Committee of Medicine Faculty of Universidad Autónoma de Nuevo León (Register number BI0002).All authors read and approved the final manuscript.The authors declare that they have no competing interests.Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Figures






References
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

