Noninvasive Magnetogastrography Detects Erythromycin-Induced Effects on the Gastric Slow Wave
- PMID: 29993499
- PMCID: PMC6411057
- DOI: 10.1109/TBME.2018.2837647
Noninvasive Magnetogastrography Detects Erythromycin-Induced Effects on the Gastric Slow Wave
Abstract
Objective: The prokinetic action of erythromycin is clinically useful under conditions associated with gastrointestinal hypomotility. Although erythromycin is known to affect the electrogastrogram, no studies have examined the effects that erythromycin has on gastric slow wave magnetic fields.
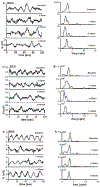
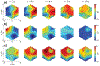
Methods: In this study, gastric slow wave activity was assessed simultaneously using noninvasive magnetogastrogram (MGG), electrogastrogram, and mucosal electromyogram recordings. Recordings were obtained for 30 min prior to and 60 min after intravenous administration of erythromycin at dosages of 3 and 6 mg/kg.
Results: MGG recordings showed significant changes in the percentage power distribution of gastric signal after infusion of both 3 and 6 mg/kg erythromycin at t = 1-5 min that persisted for t = 30-40 min after infusion. These changes agree with the changes observed in the electromyogram. We did not observe any statistically significant difference in MGG amplitude before or after injection of either 3 or 6 mg/kg erythromycin. Both 3 and 6 mg/kg erythromycin infusion showed retrograde propagation with a statistically significant decrease in slow wave propagation velocity 11-20 min after infusion. Propagation velocity started returning toward baseline values after approximately 21-30 min for the 3 mg/kg dosage and after 31-40 min for a dosage of 6 mg/kg.
Conclusion: Our results showed that the magnetic signatures were sensitive to disruptions in normal slow wave activity induced by pharmacological and prokinetic agents such as erythromycin.
Significance: This study shows that repeatable noninvasive bio-electro-magnetic techniques can objectively characterize gastric dysrhythmias and may quantify treatment efficacy in patients with functional gastric disorders.
Figures


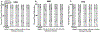
References
-
- Tomomasa T, et al., “Erythromycin Induces Migrating Motor Complex in Human Gastrointestinal-Tract”, Digestive Diseases and Sciences, vol. 31, no. 2, pp. 157–161, February, 1986. - PubMed
-
- Janssens J, et al., “Improvement of Gastric-Emptying in Diabetic Gastroparesis by Erythromycin - Preliminary Studies”, New England Journal of Medicine, vol. 322, no. 15, pp. 1028–1031, April 12, 1990. - PubMed
-
- Urbain JLC, et al., “Intravenous Erythromycin Dramatically Accelerates Gastric-Emptying in Gastroparesis Diabeticorum and Normals and Abolishes the Emptying Discrimination between Solids and Liquids”, Journal of Nuclear Medicine, vol. 31, no. 9, pp. 1490–1493, September, 1990. - PubMed
-
- Boivin MA, et al., “Erythromycin accelerates gastric emptying in a dose-response manner in healthy subjects”, Pharmacotherapy, vol. 23, no. 1, pp. 5–8, January, 2003. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

