Cyclin G and the Polycomb Repressive complexes PRC1 and PR-DUB cooperate for developmental stability
- PMID: 29995890
- PMCID: PMC6065198
- DOI: 10.1371/journal.pgen.1007498
Cyclin G and the Polycomb Repressive complexes PRC1 and PR-DUB cooperate for developmental stability
Abstract
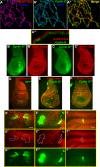
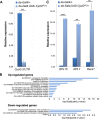
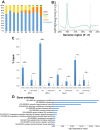
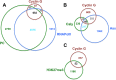
In Drosophila, ubiquitous expression of a short Cyclin G isoform generates extreme developmental noise estimated by fluctuating asymmetry (FA), providing a model to tackle developmental stability. This transcriptional cyclin interacts with chromatin regulators of the Enhancer of Trithorax and Polycomb (ETP) and Polycomb families. This led us to investigate the importance of these interactions in developmental stability. Deregulation of Cyclin G highlights an organ intrinsic control of developmental noise, linked to the ETP-interacting domain, and enhanced by mutations in genes encoding members of the Polycomb Repressive complexes PRC1 and PR-DUB. Deep-sequencing of wing imaginal discs deregulating CycG reveals that high developmental noise correlates with up-regulation of genes involved in translation and down-regulation of genes involved in energy production. Most Cyclin G direct transcriptional targets are also direct targets of PRC1 and RNAPolII in the developing wing. Altogether, our results suggest that Cyclin G, PRC1 and PR-DUB cooperate for developmental stability.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures





Similar articles
-
PR-DUB preserves Polycomb repression by preventing excessive accumulation of H2Aub1, an antagonist of chromatin compaction.Genes Dev. 2022 Oct 1;36(19-20):1046-1061. doi: 10.1101/gad.350014.122. Epub 2022 Nov 10. Genes Dev. 2022. PMID: 36357125 Free PMC article.
-
Developmental stability: a major role for cyclin G in drosophila melanogaster.PLoS Genet. 2011 Oct;7(10):e1002314. doi: 10.1371/journal.pgen.1002314. Epub 2011 Oct 6. PLoS Genet. 2011. PMID: 21998598 Free PMC article.
-
The enhancer of trithorax and polycomb corto interacts with cyclin G in Drosophila.PLoS One. 2008 Feb 20;3(2):e1658. doi: 10.1371/journal.pone.0001658. PLoS One. 2008. PMID: 18286205 Free PMC article.
-
Molecular architecture of polycomb repressive complexes.Biochem Soc Trans. 2017 Feb 8;45(1):193-205. doi: 10.1042/BST20160173. Biochem Soc Trans. 2017. PMID: 28202673 Free PMC article. Review.
-
[Maintenance of the patterns of expression of homeotic genes in the development of Drosophila melanogaster by proteins of the polycomb, trithorax, and ETP groups].Genetika. 2009 Oct;45(10):1301-18. Genetika. 2009. PMID: 19947541 Review. Russian.
Cited by
-
PRC2 promotes canalisation during endodermal differentiation.PLoS Genet. 2025 Jan 30;21(1):e1011584. doi: 10.1371/journal.pgen.1011584. eCollection 2025 Jan. PLoS Genet. 2025. PMID: 39883738 Free PMC article.
-
Mother-Fetus Immune Cross-Talk Coordinates "Extrinsic"/"Intrinsic" Embryo Gene Expression Noise and Growth Stability.Int J Mol Sci. 2022 Oct 18;23(20):12467. doi: 10.3390/ijms232012467. Int J Mol Sci. 2022. PMID: 36293324 Free PMC article.
-
Single amino-acid mutation in a Drosoph ila melanogaster ribosomal protein: An insight in uL11 transcriptional activity.PLoS One. 2022 Aug 18;17(8):e0273198. doi: 10.1371/journal.pone.0273198. eCollection 2022. PLoS One. 2022. PMID: 35981051 Free PMC article.
-
Systemic signalling and local effectors in developmental stability, body symmetry, and size.Cell Stress. 2018 Nov 13;2(12):340-361. doi: 10.15698/cst2018.12.167. Cell Stress. 2018. PMID: 31225459 Free PMC article. Review.
References
-
- Palmer RA (1994) Fluctuating asymmetry analysis: a primer In: Developmental Instability: Its Origins and Evolutionary Implications. Markow T. A. Netherlands, Kluwer Academic Publishers; pp. 335–364.
-
- Nijhout F, Davidowitz G (2003) Developmental Perspectives on Phenotypic Variation, Canalization, and Fluctuating Asymmetry In: Polak M, editors. Developmental instability, causes and consequences. New York: Oxford University Press; pp. 3–13.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

