Effects of lyophilization and storage temperature on Wuchereria bancrofti antigen sensitivity and stability
- PMID: 29996898
- PMCID: PMC6042357
- DOI: 10.1186/s13104-018-3586-0
Effects of lyophilization and storage temperature on Wuchereria bancrofti antigen sensitivity and stability
Abstract
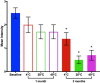
Objective: Antigen-based rapid diagnostic tests for Lymphatic filariasis (LF) do not come with external quality control (QC) materials, and research and disease control programmes rely on stored positive samples. This study was undertaken to evaluate the use of lyophilized Wuchereria bancrofti antigen positive plasma samples to serve as QC materials for LF diagnostic tests. 10 well characterized W. bancrofti positive samples were lyophilized and stored at 4, 28 and 40 °C. The samples were evaluated using the Alere Filariasis Test Strips before lyophilization, and after 1 and 3 months of storage. The sensitivity and stability of the lyophilized samples were evaluated.
Results: The results revealed a loss of sensitivity and stability with increasing temperature and duration of storage. The results are further discussed in terms of the use of dried blood spot (DBS) in diagnostic studies on LF, and the need for thoughtful DBS preparation and storage.
Keywords: Dried blood spots; Freeze-drying; Lyophilization; Wuchereria bancrofti antigen.
Figures
Similar articles
-
Rapid Wuchereria bancrofti-specific antigen Wb123-based IgG4 immunoassays as tools for surveillance following mass drug administration programs on lymphatic filariasis.Clin Vaccine Immunol. 2013 Aug;20(8):1155-61. doi: 10.1128/CVI.00252-13. Epub 2013 Jun 5. Clin Vaccine Immunol. 2013. PMID: 23740923 Free PMC article.
-
Evaluation of in vitro released Wuchereria bancrofti third stage larval antigens for detection of Bancroftian filariasis.Trop Med Parasitol. 1990 Mar;41(1):71-2. Trop Med Parasitol. 1990. PMID: 2187228
-
Evaluation of Og4C3 antigen ELISA as a tool for detection of bancroftian filariasis under lymphatic filariasis elimination programme.J Commun Dis. 2007 Jun;39(2):75-84. J Commun Dis. 2007. PMID: 18338684
-
Detection and diagnostic utility of in vitro and in vivo released antigens in bancroftian filariasis.J Commun Dis. 1986 Dec;18(4):261-6. J Commun Dis. 1986. PMID: 3309030 Review. No abstract available.
-
Recombinant antigens used as diagnostic tools for lymphatic filariasis.Parasit Vectors. 2021 Sep 15;14(1):474. doi: 10.1186/s13071-021-04980-3. Parasit Vectors. 2021. PMID: 34526120 Free PMC article. Review.
Cited by
-
Freeze-drying revolution: unleashing the potential of lyophilization in advancing drug delivery systems.Drug Deliv Transl Res. 2024 May;14(5):1111-1153. doi: 10.1007/s13346-023-01477-7. Epub 2023 Nov 20. Drug Deliv Transl Res. 2024. PMID: 37985541 Review.
-
Storage conditions affect the composition of the lyophilized secretome of multipotent mesenchymal stromal cells.Sci Rep. 2024 May 3;14(1):10243. doi: 10.1038/s41598-024-60787-z. Sci Rep. 2024. PMID: 38702388 Free PMC article.
References
-
- WHO Global programme to eliminate lymphatic filariasis: progress report, 2016. Wkly Epidemiol Rec. 2017;92:589–608. - PubMed
-
- WHO . Monitoring and epidemiological assessment of mass drug administration in the global programme to eliminate lymphatic filariasis: a manual for national elimination programmes. WHO/HTM/NTD/PCT/2011. Geneva: WHO; 2011. pp. 1–79.
-
- WHO . Guideline—alternative mass drug administration regimens to eliminate lymphatic filariasis. WHO/HTM/NTD/PCT/2017. Geneva: WHO; 2017. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources