SPOP promotes ATF2 ubiquitination and degradation to suppress prostate cancer progression
- PMID: 29996942
- PMCID: PMC6042370
- DOI: 10.1186/s13046-018-0809-0
SPOP promotes ATF2 ubiquitination and degradation to suppress prostate cancer progression
Abstract
Background: Next-generation sequencing of the exome and genome of prostate cancers has identified numerous genetic alterations. SPOP (Speckle-type POZ Protein) is one of the most frequently mutated genes in primary prostate cancer, suggesting that SPOP may be a potential driver of prostate cancer. The aim of this work was to investigate how SPOP mutations contribute to prostate cancer development and progression.
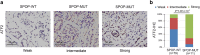
Methods: To identify molecular mediators of the tumor suppressive function of SPOP, we performed a yeast two-hybrid screen in a HeLa cDNA library using the full-length SPOP as bait. Immunoprecipitation and Western Blotting were used to analyze the interaction between SPOP and ATF2. Cell migration and invasion were determined by Transwell assays. Immunohistochemistry were used to analyze protein levels in patients' tumor samples.
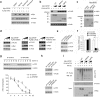
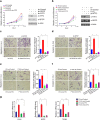
Results: Here we identified ATF2 as a bona fide substrate of the SPOP-CUL3-RBX1 E3 ubiquitin ligase complex. SPOP recognizes multiple Ser/Thr (S/T)-rich degrons in ATF2 and triggers ATF2 degradation via the ubiquitin-proteasome pathway. Strikingly, prostate cancer-associated mutants of SPOP are defective in promoting ATF2 degradation in prostate cancer cells and contribute to facilitating prostate cancer cell proliferation, migration and invasion.
Conclusion: SPOP promotes ATF2 ubiquitination and degradation, and ATF2 is an important mediator of SPOP inactivation-induced cell proliferation, migration and invasion.
Keywords: ATF2; Prostate cancer; Proteasomal degradation; SPOP; Ubiquitination.
Conflict of interest statement
Ethics approval and consent to participate
The study was approved by the ethics committee of the Fudan University Shanghai Cancer Center and signed informed consent was obtained from each patient.
Consent for publication
All the authors have read and approved the manuscript.
Competing interests
The authors declare that they have no competing interests.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Figures



Similar articles
-
Destruction of DDIT3/CHOP protein by wild-type SPOP but not prostate cancer-associated mutants.Hum Mutat. 2014 Sep;35(9):1142-51. doi: 10.1002/humu.22614. Epub 2014 Jul 23. Hum Mutat. 2014. PMID: 24990631
-
Dysregulation of INF2-mediated mitochondrial fission in SPOP-mutated prostate cancer.PLoS Genet. 2017 Apr 27;13(4):e1006748. doi: 10.1371/journal.pgen.1006748. eCollection 2017 Apr. PLoS Genet. 2017. PMID: 28448495 Free PMC article.
-
Endometrial cancer-associated mutants of SPOP are defective in regulating estrogen receptor-α protein turnover.Cell Death Dis. 2015 Mar 12;6(3):e1687. doi: 10.1038/cddis.2015.47. Cell Death Dis. 2015. PMID: 25766326 Free PMC article.
-
The ubiquitin ligase adaptor SPOP in cancer.FEBS J. 2019 Oct;286(20):3946-3958. doi: 10.1111/febs.15056. Epub 2019 Sep 18. FEBS J. 2019. PMID: 31495053 Free PMC article. Review.
-
The emerging role of speckle-type POZ protein (SPOP) in cancer development.Drug Discov Today. 2014 Sep;19(9):1498-502. doi: 10.1016/j.drudis.2014.07.009. Epub 2014 Jul 21. Drug Discov Today. 2014. PMID: 25058385 Free PMC article. Review.
Cited by
-
Detection of disease-causing mutations in prostate cancer by NGS sequencing.Cell Biol Int. 2022 Jul;46(7):1047-1061. doi: 10.1002/cbin.11803. Epub 2022 Apr 6. Cell Biol Int. 2022. PMID: 35347810 Free PMC article.
-
The emerging role of SPOP protein in tumorigenesis and cancer therapy.Mol Cancer. 2020 Jan 4;19(1):2. doi: 10.1186/s12943-019-1124-x. Mol Cancer. 2020. PMID: 31901237 Free PMC article. Review.
-
The diverse roles of SPOP in prostate cancer and kidney cancer.Nat Rev Urol. 2020 Jun;17(6):339-350. doi: 10.1038/s41585-020-0314-z. Epub 2020 Apr 30. Nat Rev Urol. 2020. PMID: 32355326 Review.
-
Phytochemical profiling and anticancer potential of pistachio wastes against MCF-7 breast cancer cells: a metabolic and apoptotic pathway analysis.BMC Complement Med Ther. 2025 Jul 17;25(1):275. doi: 10.1186/s12906-025-04963-2. BMC Complement Med Ther. 2025. PMID: 40676557 Free PMC article.
-
Function, mechanism and drug discovery of ubiquitin and ubiquitin-like modification with multiomics profiling for cancer therapy.Acta Pharm Sin B. 2023 Nov;13(11):4341-4372. doi: 10.1016/j.apsb.2023.07.019. Epub 2023 Jul 22. Acta Pharm Sin B. 2023. PMID: 37969742 Free PMC article. Review.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

