Improving the Catalytic Performance of Keggin [PW12O40]3- for Oxidative Desulfurization: Ionic Liquids versus SBA-15 Composite
- PMID: 30002316
- PMCID: PMC6073681
- DOI: 10.3390/ma11071196
Improving the Catalytic Performance of Keggin [PW12O40]3- for Oxidative Desulfurization: Ionic Liquids versus SBA-15 Composite
Abstract
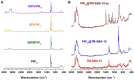
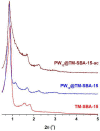
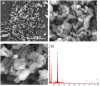
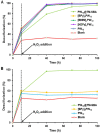
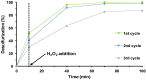
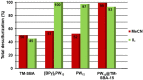
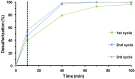
Different methodologies were used to increase the oxidative desulfurization efficiency of the Keggin phosphotungstate [PW12O40]3- (PW12). One possibility was to replace the acid proton by three different ionic liquid cations, forming the novel hybrid polyoxometalates: [BMIM]₃PW12 (BMIM as 1-butyl-3-methylimidazolium), [BPy]₃PW12 (BPy as 1-butylpyridinium) and [HDPy]₃PW12 (HDPy as hexadecylpyridinium. These hybrid Keggin compounds showed high oxidative desulfurization efficiency in the presence of [BMIM]PF₆ solvent, achieving complete desulfurization of multicomponent model diesel (2000 ppm of S) after only 1 h, using a low excess of oxidant (H₂O₂/S = 8) at 70 °C. However, their stability and activity showed some weakness in continuous reused oxidative desulfurization cycles. An improvement of stability in continuous reused cycles was reached by the immobilization of the Keggin polyanion in a strategic positively-charged functionalized-SBA-15 support. The PW12@TM⁻SBA-15 composite (TM is the trimethylammonium functional group) presented similar oxidative desulfurization efficiency to the homogeneous IL⁻PW12 compounds, having the advantage of a high recycling capability in continuous cycles, increasing its activity from the first to the consecutive cycles. Therefore, the oxidative desulfurization system catalyzed by the Keggin-type composite has high performance under sustainable operational conditions, avoids waste production during recycling and allows catalyst recovery.
Keywords: benzothiophene derivatives; functionalized-SBA-15; hydrogen peroxide; ionic liquids polyoxometalates; sustainable oxidative desulfurization.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
Mesoporous Silica vs. Organosilica Composites to Desulfurize Diesel.Front Chem. 2019 Nov 14;7:756. doi: 10.3389/fchem.2019.00756. eCollection 2019. Front Chem. 2019. PMID: 31799236 Free PMC article.
-
Effective Combination of the Metal Centers in MOF-Based Materials toward Sustainable Oxidation Catalysts.Materials (Basel). 2023 Apr 16;16(8):3133. doi: 10.3390/ma16083133. Materials (Basel). 2023. PMID: 37109968 Free PMC article.
-
Ionic Liquid-Based Polyoxometalate Incorporated at ZIF-8: A Sustainable Catalyst to Combine Desulfurization and Denitrogenation Processes.Molecules. 2022 Mar 5;27(5):1711. doi: 10.3390/molecules27051711. Molecules. 2022. PMID: 35268812 Free PMC article.
-
Deep Oxidative Desulfurization of Fuels in the Presence of Brönsted Acidic Polyoxometalate-Based Ionic Liquids.Molecules. 2020 Jan 26;25(3):536. doi: 10.3390/molecules25030536. Molecules. 2020. PMID: 31991874 Free PMC article.
-
A current overview of the oxidative desulfurization of fuels utilizing heat and solar light: from materials design to catalysis for clean energy.Nanoscale Horiz. 2021 Jul 26;6(8):588-633. doi: 10.1039/d1nh00127b. Nanoscale Horiz. 2021. PMID: 34018529 Review.
Cited by
-
Studies on nitrate acid based imidazolium ionic liquids: synthesis and application in electrochemical desulfurization of oil.RSC Adv. 2025 Jun 2;15(23):18283-18291. doi: 10.1039/d5ra01724f. eCollection 2025 May 29. RSC Adv. 2025. PMID: 40458431 Free PMC article.
-
A Novel Ternary Catalyst PW4@MOF-808@SBA-15 for Deep Extraction Oxidation Desulfurization of Model Diesel.Molecules. 2024 Sep 6;29(17):4230. doi: 10.3390/molecules29174230. Molecules. 2024. PMID: 39275078 Free PMC article.
-
Solvent-Free Desulfurization System to Produce Low-Sulfur Diesel Using Hybrid Monovacant Keggin-Type Catalyst.Molecules. 2020 Oct 27;25(21):4961. doi: 10.3390/molecules25214961. Molecules. 2020. PMID: 33120916 Free PMC article.
-
Heteropolyacid Ionic Liquid-Based MCF: An Efficient Heterogeneous Catalyst for Oxidative Desulfurization of Fuel.Materials (Basel). 2023 Apr 18;16(8):3195. doi: 10.3390/ma16083195. Materials (Basel). 2023. PMID: 37110030 Free PMC article.
-
Mesoporous Silica vs. Organosilica Composites to Desulfurize Diesel.Front Chem. 2019 Nov 14;7:756. doi: 10.3389/fchem.2019.00756. eCollection 2019. Front Chem. 2019. PMID: 31799236 Free PMC article.
References
-
- Samokhvalov A., Tatarchuk B.J. Review of Experimental Characterization of Active Sites and Determination of Molecular Mechanisms of Adsorption, Desorption and Regeneration of the Deep and Ultradeep Desulfurization Sorbents for Liquid Fuels. Catal. Rev. 2010;52:381–410. doi: 10.1080/01614940.2010.498749. - DOI
-
- Torres-García E., Galano A., Rodriguez-Gattorno G. Oxidative desulfurization (ODS) of organosulfur compounds catalyzed by peroxo-metallate complexes of WOx–ZrO2: Thermochemical, structural, and reactivity indexes analyses. J. Catal. 2011;282:201–208. doi: 10.1016/j.jcat.2011.06.010. - DOI
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

