CORL Expression and Function in Insulin Producing Neurons Reversibly Influences Adult Longevity in Drosophila
- PMID: 30006413
- PMCID: PMC6118311
- DOI: 10.1534/g3.118.200572
CORL Expression and Function in Insulin Producing Neurons Reversibly Influences Adult Longevity in Drosophila
Abstract
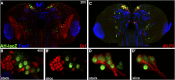
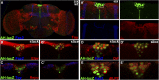
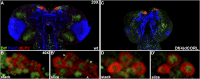
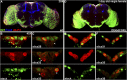
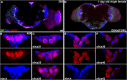
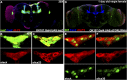
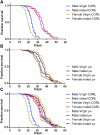
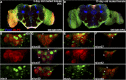
CORL proteins (known as SKOR in mice, Fussel in humans and fussel in Flybase) are a family of CNS specific proteins related to Sno/Ski oncogenes. Their developmental and adult roles are largely unknown. A Drosophila CORL (dCORL) reporter gene is expressed in all Drosophila insulin-like peptide 2 (dILP2) neurons of the pars intercerebralis (PI) of the larval and adult brain. The transcription factor Drifter is also expressed in the PI in a subset of dCORL and dILP2 expressing neurons and in several non-dILP2 neurons. dCORL mutant virgin adult brains are missing all dILP2 neurons that do not also express Drifter. This phenotype is also seen when expressing dCORL-RNAi in neurosecretory cells of the PI. dCORL mutant virgin adults of both sexes have a significantly shorter lifespan than their parental strain. This longevity defect is completely reversed by mating (lifespan increases over 50% for males and females). Analyses of dCORL mutant mated adult brains revealed a complete rescue of dILP2 neurons without Drifter. Taken together, the data suggest that dCORL participates in a neural network connecting the insulin signaling pathway, longevity and mating. The conserved sequence and CNS specificity of all CORL proteins imply that this network may be operating in mammals.
Keywords: Drifter; Fussel/SKOR; dILP2; lifespan extension; pars intercerebralis.
Copyright © 2018 Tran et al.
Figures
Similar articles
-
Transgenic Analyses in Drosophila Reveal That mCORL1 Is Functionally Distinct from mCORL2 and dCORL.G3 (Bethesda). 2019 Nov 5;9(11):3781-3789. doi: 10.1534/g3.119.400647. G3 (Bethesda). 2019. PMID: 31530634 Free PMC article.
-
Adult Movement Defects Associated with a CORL Mutation in Drosophila Display Behavioral Plasticity.G3 (Bethesda). 2020 May 4;10(5):1697-1706. doi: 10.1534/g3.120.400648. G3 (Bethesda). 2020. PMID: 32161085 Free PMC article.
-
CORL Expression in the Drosophila Central Nervous System Is Regulated by Stage Specific Interactions of Intertwined Activators and Repressors.G3 (Bethesda). 2018 Jul 2;8(7):2527-2536. doi: 10.1534/g3.118.200282. G3 (Bethesda). 2018. PMID: 29848623 Free PMC article.
-
[Neuroendocrine system in Drosophila melanogaster lifespan control].Adv Gerontol. 2010;23(4):518-26. Adv Gerontol. 2010. PMID: 21510072 Review. Russian.
-
Functional implications of Drosophila insulin-like peptides in metabolism, aging, and dietary restriction.Front Physiol. 2013 Oct 16;4:288. doi: 10.3389/fphys.2013.00288. Front Physiol. 2013. PMID: 24137131 Free PMC article. Review.
Cited by
-
New resources for the Drosophila 4th chromosome: FRT101F enabled mitotic clones and Bloom syndrome helicase enabled meiotic recombination.G3 (Bethesda). 2022 Apr 4;12(4):jkac019. doi: 10.1093/g3journal/jkac019. G3 (Bethesda). 2022. PMID: 35084488 Free PMC article.
-
Transgenic Analyses in Drosophila Reveal That mCORL1 Is Functionally Distinct from mCORL2 and dCORL.G3 (Bethesda). 2019 Nov 5;9(11):3781-3789. doi: 10.1534/g3.119.400647. G3 (Bethesda). 2019. PMID: 31530634 Free PMC article.
-
dSmad2 differentially regulates dILP2 and dILP5 in insulin producing and circadian pacemaker cells in unmated adult females.PLoS One. 2023 Jan 23;18(1):e0280529. doi: 10.1371/journal.pone.0280529. eCollection 2023. PLoS One. 2023. PMID: 36689407 Free PMC article.
-
Adult Movement Defects Associated with a CORL Mutation in Drosophila Display Behavioral Plasticity.G3 (Bethesda). 2020 May 4;10(5):1697-1706. doi: 10.1534/g3.120.400648. G3 (Bethesda). 2020. PMID: 32161085 Free PMC article.
-
The Drosophila fussel gene is required for bitter gustatory neuron differentiation acting within an Rpd3 dependent chromatin modifying complex.PLoS Genet. 2019 Feb 7;15(2):e1007940. doi: 10.1371/journal.pgen.1007940. eCollection 2019 Feb. PLoS Genet. 2019. PMID: 30730884 Free PMC article.
References
-
- Chapman T., Liddle L., Kalb J., Wolfner M., Partridge L., 1995. Cost of mating in Drosophila melanogaster females is mediated by male accessory gland products. Nature 373: 241–244. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous
