Enhanced motility and proliferation by miR-10b/FUT8/p-AKT axis in breast cancer cells
- PMID: 30008906
- PMCID: PMC6036446
- DOI: 10.3892/ol.2018.8891
Enhanced motility and proliferation by miR-10b/FUT8/p-AKT axis in breast cancer cells
Abstract
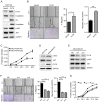
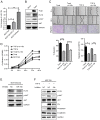
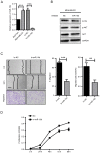
Upregulation of microRNA (miR)-10b has been confirmed in multiple types of cancer, however, the role of miR-10b in glycosylation remains unclear. Protein core-fucosylation is an important N-linked glycosylation modification and serves important roles in cancer progression. In a previous study, a glycogene array was applied to profile the alterations of glycogene expression in miR-10b-overexpressed MCF10A cells. Notably, fucosyltranferase 8 (FUT8), which is responsible for the addition of core-fucose to N-glycan, was significantly upregulated by miR-10b. In the present study, increased motility and proliferation were observed in miR-10b-overexpressed MCF10A cells. To assess the mechanism involved, the role of FUT8 in MCF10A cells was studied and it was confirmed that miR-10b promotes motility and proliferation by regulating FUT8 and activating the protein kinase B (AKT) signaling pathway. Consistent with the aforementioned result, decreased motility and proliferation were detected when miR-10b expression was inhibited in MDA-MB-231 cells, transforming growth factor-β-induced and Twist-overexpressed MCF10A cells. To conclude, the findings from the present study indicate that miR-10b promotes motility and proliferation by increasing FUT8 and activating AKT in breast cancer cells.
Keywords: breast cancer; fucosyltranferase 8; microRNA-10b; motility; phosphorylated protein kinase B; proliferation.
Figures

Similar articles
-
FUT8 promotes breast cancer cell invasiveness by remodeling TGF-β receptor core fucosylation.Breast Cancer Res. 2017 Oct 5;19(1):111. doi: 10.1186/s13058-017-0904-8. Breast Cancer Res. 2017. PMID: 28982386 Free PMC article.
-
Targeting of syndecan-1 by microRNA miR-10b promotes breast cancer cell motility and invasiveness via a Rho-GTPase- and E-cadherin-dependent mechanism.Int J Cancer. 2012 Sep 15;131(6):E884-96. doi: 10.1002/ijc.27629. Epub 2012 May 30. Int J Cancer. 2012. PMID: 22573479
-
Comprehensive N-glycan profiles of hepatocellular carcinoma reveal association of fucosylation with tumor progression and regulation of FUT8 by microRNAs.Oncotarget. 2016 Sep 20;7(38):61199-61214. doi: 10.18632/oncotarget.11284. Oncotarget. 2016. PMID: 27533464 Free PMC article.
-
Estrogen receptor beta as epigenetic mediator of miR-10b and miR-145 in mammary cancer.Matrix Biol. 2017 Dec;64:94-111. doi: 10.1016/j.matbio.2017.08.002. Epub 2017 Aug 8. Matrix Biol. 2017. PMID: 28797712
-
FUT8 and Protein Core Fucosylation in Tumours: From Diagnosis to Treatment.J Cancer. 2021 May 13;12(13):4109-4120. doi: 10.7150/jca.58268. eCollection 2021. J Cancer. 2021. PMID: 34093814 Free PMC article. Review.
Cited by
-
Hexokinase 2 promoted cell motility and proliferation by activating Akt1/p-Akt1 in human ovarian cancer cells.J Ovarian Res. 2022 Aug 11;15(1):92. doi: 10.1186/s13048-022-01027-8. J Ovarian Res. 2022. PMID: 35953860 Free PMC article.
-
The Role of Epithelial Mesenchymal Transition (EMT) in Pathogenesis of Cardiotoxicity: Diagnostic & Prognostic Approach.Mol Biotechnol. 2023 Sep;65(9):1403-1413. doi: 10.1007/s12033-023-00697-z. Epub 2023 Feb 27. Mol Biotechnol. 2023. PMID: 36847962 Review.
-
Defining the altered glycoproteomic space of the early secretory pathway by class I mannosidase pharmacological inhibition.Front Mol Biosci. 2023 Jan 9;9:1064868. doi: 10.3389/fmolb.2022.1064868. eCollection 2022. Front Mol Biosci. 2023. PMID: 36699698 Free PMC article.
-
Fucosyltransferase 8 regulation and breast cancer suppression by transcription factor activator protein 2γ.Cancer Sci. 2021 Aug;112(8):3190-3204. doi: 10.1111/cas.14987. Epub 2021 Jun 12. Cancer Sci. 2021. PMID: 34036684 Free PMC article.
-
Targeting Post-Translational Modifications to Improve Combinatorial Therapies in Breast Cancer: The Role of Fucosylation.Cells. 2023 Mar 8;12(6):840. doi: 10.3390/cells12060840. Cells. 2023. PMID: 36980181 Free PMC article. Review.
References
-
- Peart O. Metastatic breast cancer. Radiol Technol. 2017;88:519M–539M. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous
