Drosophila species learn dialects through communal living
- PMID: 30024883
- PMCID: PMC6053138
- DOI: 10.1371/journal.pgen.1007430
Drosophila species learn dialects through communal living
Erratum in
-
Correction: Drosophila species learn dialects through communal living.PLoS Genet. 2018 Nov 27;14(11):e1007825. doi: 10.1371/journal.pgen.1007825. eCollection 2018 Nov. PLoS Genet. 2018. PMID: 30481167 Free PMC article.
Abstract
Many species are able to share information about their environment by communicating through auditory, visual, and olfactory cues. In Drosophila melanogaster, exposure to parasitoid wasps leads to a decline in egg laying, and exposed females communicate this threat to naïve flies, which also depress egg laying. We find that species across the genus Drosophila respond to wasps by egg laying reduction, activate cleaved caspase in oocytes, and communicate the presence of wasps to naïve individuals. Communication within a species and between closely related species is efficient, while more distantly related species exhibit partial communication. Remarkably, partial communication between some species is enhanced after a cohabitation period that requires exchange of visual and olfactory signals. This interspecies "dialect learning" requires neuronal cAMP signaling in the mushroom body, suggesting neuronal plasticity facilitates dialect learning and memory. These observations establish Drosophila as genetic models for interspecies social communication and evolution of dialects.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures
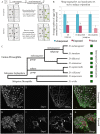
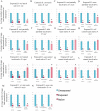
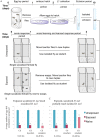





Comment in
-
Multiculturalism is good for flies, too.PLoS Genet. 2018 Jul 19;14(7):e1007480. doi: 10.1371/journal.pgen.1007480. eCollection 2018 Jul. PLoS Genet. 2018. PMID: 30024880 Free PMC article. No abstract available.
References
-
- Gould JL. Honey bee communication. Nature 1974.
-
- Wenner AM. Sound production during the waggle dance of the honey bee. Anim Behav 1962;10(1):79–95.
-
- Winston ML. The biology of the honey bee.: harvard university press; 1991.
-
- Goodale E, Beauchamp G, Magrath RD, Nieh JC, Ruxton GD. Interspecific information transfer influences animal community structure. Trends in ecology & evolution 2010;25(6):354–361. - PubMed
-
- Westrip JR, Bell MB. Breaking down the species boundaries: selective pressures behind interspecific communication in vertebrates. Ethology 2015;121(8):725–732.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

