Quantification of anti-parasite and anti-disease immunity to malaria as a function of age and exposure
- PMID: 30044224
- PMCID: PMC6103767
- DOI: 10.7554/eLife.35832
Quantification of anti-parasite and anti-disease immunity to malaria as a function of age and exposure
Abstract
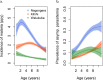
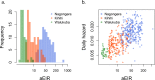
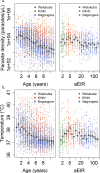
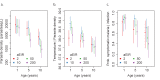
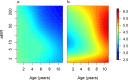
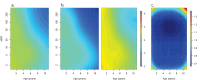
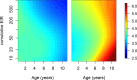
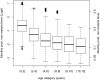
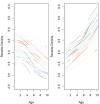
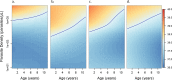
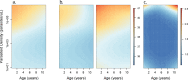
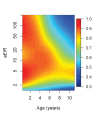
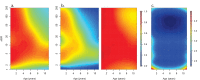
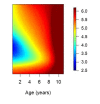
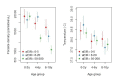
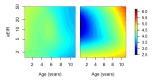
Fundamental gaps remain in our understanding of how immunity to malaria develops. We used detailed clinical and entomological data from parallel cohort studies conducted across the malaria transmission spectrum in Uganda to quantify the development of immunity against symptomatic P. falciparum as a function of age and transmission intensity. We focus on: anti-parasite immunity (i.e. ability to control parasite densities) and anti-disease immunity (i.e. ability to tolerate higher parasite densities without fever). Our findings suggest a strong effect of age on both types of immunity, not explained by cumulative-exposure. They also show an independent effect of exposure, where children living in moderate/high transmission settings develop immunity faster as transmission increases. Surprisingly, children in the lowest transmission setting appear to develop immunity more efficiently than those living in moderate transmission settings. Anti-parasite and anti-disease immunity develop in parallel, reducing the probability of experiencing symptomatic malaria upon each subsequent P. falciparum infection.
Keywords: P. falciparum; anti-disease immunity; anti-parasite immunity; epidemiology; global health; immunity; malaria.
© 2018, Rodriguez-Barraquer et al.
Conflict of interest statement
IR, EA, PJ, MK, PR, JR, GD, JN, SS, MK, CD, IS, DS, BG No competing interests declared
Figures

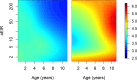

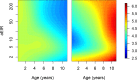
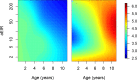

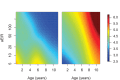





Comment in
-
Age, exposure and immunity.Elife. 2018 Aug 21;7:e40150. doi: 10.7554/eLife.40150. Elife. 2018. PMID: 30129437 Free PMC article.
References
-
- Bhatt S, Weiss DJ, Cameron E, Bisanzio D, Mappin B, Dalrymple U, Battle K, Moyes CL, Henry A, Eckhoff PA, Wenger EA, Briët O, Penny MA, Smith TA, Bennett A, Yukich J, Eisele TP, Griffin JT, Fergus CA, Lynch M, Lindgren F, Cohen JM, Murray CLJ, Smith DL, Hay SI, Cibulskis RE, Gething PW. The effect of malaria control on Plasmodium falciparum in Africa between 2000 and 2015. Nature. 2015;526:207–211. doi: 10.1038/nature15535. - DOI - PMC - PubMed
-
- Carneiro I, Roca-Feltrer A, Griffin JT, Smith L, Tanner M, Schellenberg JA, Greenwood B, Schellenberg D. Age-patterns of malaria vary with severity, transmission intensity and seasonality in sub-Saharan Africa: a systematic review and pooled analysis. PLoS One. 2010;5:e8988. doi: 10.1371/journal.pone.0008988. - DOI - PMC - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

