Lung epithelial cell-derived IL-25 negatively regulates LPS-induced exosome release from macrophages
- PMID: 30056803
- PMCID: PMC6065058
- DOI: 10.1186/s40779-018-0173-6
Lung epithelial cell-derived IL-25 negatively regulates LPS-induced exosome release from macrophages
Abstract
Background: Acute lung injury (ALI) is a major component of multiple organ dysfunction syndrome (MODS) following pulmonary and systemic infection. Alveolar macrophages (AMϕ) are at the center of ALI pathogenesis. Emerging evidence has shown that cell-cell interactions in the lungs play an important regulatory role in the development of acute lung inflammation. However, the underneath mechanisms remain poorly addressed. In this study, we explore a novel function of lung epithelial cells (LEPCs) in regulating the release of exosomes from AMϕ following LPS stimulation.
Methods: For the in vivo experiments, C57BL/6 wildtype (WT) mice were treated with lipopolysaccharide (LPS) (2 mg/kg B.W.) in 0.2 ml of saline via intratracheal aerosol administration. Bronchoalveolar lavage fluid was collected at 0-24 h after LPS treatment, and exosomes derived from AMϕ were measured. For the in vitro studies, LEPCs and bone marrow-derived Mϕ (BMDM) were isolated from WT or TLR4-/- mice and were then cocultured in the Transwell™ system. After coculture for 0-24 h, the BMDM and supernatant were harvested for the measurement of exosomes and cytokines.
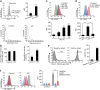
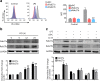
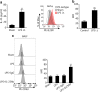
Results: We demonstrate that LPS induces macrophages (Mϕ) to release exosomes, which are then internalized by neighboring Mϕ to promote TNF-α expression. The secreted interleukin (IL)-25 from LEPCs downregulates Rab27a and Rab27b expression in Mϕ, resulting in suppressed exosome release and thereby attenuating exosome-induced TNF-α expression and secretion.
Conclusion: These findings reveal a previously unidentified crosstalk pathway between LEPCs and Mϕ that negatively regulates the inflammatory responses of Mϕ to LPS. Modulating IL-25 signaling and targeting exosome release may present a new therapeutic strategy for the treatment of ALI.
Keywords: Acute lung injury; Multiple organ failure; Rab27; Sepsis.
Conflict of interest statement
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Figures


References
-
- Xiang M, Yin L, Li Y, Xiao G, Vodovotz Y, Billiar TR, et al. Hemorrhagic shock activates lung endothelial reduced nicotinamide adenine dinucleotide phosphate (NADPH) oxidase via neutrophil NADPH oxidase. Am J Respir Cell Mol Biol. 2011;44(3):333–340. doi: 10.1165/rcmb.2009-0408OC. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- 1I01BX002729/U.S. Department of Veterans Affairs/International
- R01 HL076179/HL/NHLBI NIH HHS/United States
- R01GM102146/GM/NIGMS NIH HHS/United States
- R01 HL079669/HL/NHLBI NIH HHS/United States
- R01 HL128297/HL/NHLBI NIH HHS/United States
- R01-HL-079669/HL/NHLBI NIH HHS/United States
- R01 GM102146/GM/NIGMS NIH HHS/United States
- R01HL076179-09/HL/NHLBI NIH HHS/United States
- I01 BX002729/BX/BLRD VA/United States
- R56 HL123882/HL/NHLBI NIH HHS/United States
- 81470262/National Natural Science Foundation of China/International
- IK6 BX004211/BX/BLRD VA/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources

