Ex vivo anti-inflammatory effects of probiotics for periodontal health
- PMID: 30057719
- PMCID: PMC6060379
- DOI: 10.1080/20002297.2018.1502027
Ex vivo anti-inflammatory effects of probiotics for periodontal health
Abstract
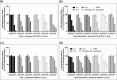
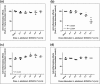
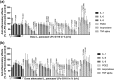
Background: Probiotic bacteria with anti-inflammatory properties have the potential to be of therapeutic benefit in gingivitis. Objective: To evaluate the effects of potential probiotic strains on inflammatory mediators involved in early gingivitis using an ex vivo inflammation model. Methods: Strains were screened in viable and attenuated forms for effects on bacterial lipopolysaccharide (LPS)-stimulated release of interleukins (IL)-1β, -6 and -8, tumor necrosis factor-α, prostaglandin E2 and 8-isoprostane from human primary monocytes, and then, if anti-inflammatory effects were shown, on IL-1β-stimulated release of inflammatory mediators from primary gingival fibroblasts. Lead strains were evaluated for optimal dosing, batch-to-batch variation and functional consistency in toothpaste. Results: Twenty-one of 73 strains showed anti-inflammatory effects in monocytes; of which, seven showed effects in both viable and attenuated forms. Seven of 14 strains showed effects in fibroblasts. Strains Lactobacillus paracasei LPc-G110(SYBIO-15) and Lactobacillus plantarum GOS42(SYBIO-41) induced statistically significant dose-dependent reductions in the release of multiple inflammatory mediators from monocytes, which were consistent across batches. Viable L. paracasei LPc-G110 tooth paste significantly reduced IL-6, IL-8 and prostaglandin E2 release from monocytes versus placebo. Conclusion: Strains L. paracasei LPc-G110 and L. plantarum GOS42 have potential for use as probiotics in oral care products to reduce gingival inflammation.
Keywords: Gingivitis; bacteria; biofilms; dental plaque; inflammation; oral hygiene; probiotics.
Figures


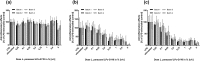


References
-
- Loe H, Theilade E, Jensen SB.. Experimental gingivitis in man. J Periodontol. 1965;36:177–10. PubMed PMID: 14296927; eng. - PubMed
-
- Madianos PN, Bobetsis YA, Kinane DF. Generation of inflammatory stimuli: how bacteria set up inflammatory responses in the gingiva. J Clin Periodontol. 2005;32 Suppl 6:57–71. PubMed PMID: 16128830; eng. - PubMed
-
- Scannapieco FA. Periodontal inflammation: from gingivitis to systemic disease? Compend Contin Educ Dent. 2004;25(7 Suppl 1): 16–25. PubMed PMID: 15645883; eng. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
