Mechanistic Insights into CpG DNA and IL-15 Synergy in Promoting B Cell Chronic Lymphocytic Leukemia Clonal Expansion
- PMID: 30068596
- PMCID: PMC6103916
- DOI: 10.4049/jimmunol.1800591
Mechanistic Insights into CpG DNA and IL-15 Synergy in Promoting B Cell Chronic Lymphocytic Leukemia Clonal Expansion
Abstract
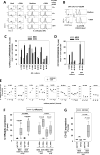
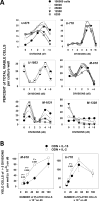
Malignant cell growth within patients with B cell chronic lymphocytic leukemia (B-CLL) is largely restricted to lymphoid tissues, particularly lymph nodes. The recent in vitro finding that TLR-9 ligand (oligodeoxynucleotide [ODN]) and IL-15 exhibit strong synergy in promoting B-CLL growth may be particularly relevant to growth in these sites. This study shows IL-15-producing cells are prevalent within B-CLL-infiltrated lymph nodes and, using purified B-CLL cells from blood, investigates the mechanism for ODN and IL-15 synergy in driving B-CLL growth. ODN boosts baseline levels of phospho-RelA(S529) in B-CLL and promotes NF-κB-driven increases in IL15RA and IL2RB mRNA, followed by elevated IL-15Rα and IL-2/IL-15Rβ (CD122) protein. IL-15→CD122 signaling during a critical interval, 20 to 36-48 h following initial ODN exposure, is required for optimal induction of the cycling process. Furthermore, experiments with neutralizing anti-IL-15 and anti-CD122 mAbs indicate that clonal expansion requires continued IL-15/CD122 signaling during cycling. The latter is consistent with evidence of heightened IL2RB mRNA in the fraction of recently proliferated B-CLL cells within patient peripheral blood. Compromised ODN+IL-15 growth with limited cell density is consistent with a role for upregulated IL-15Rα in facilitating homotypic trans IL-15 signaling, although there may be other explanations. Together, the findings show that ODN and IL-15 elicit temporally distinct signals that function in a coordinated manner to drive B-CLL clonal expansion.
Copyright © 2018 by The American Association of Immunologists, Inc.
Conflict of interest statement
The authors have no financial conflicts of interest.
Figures





Similar articles
-
Mechanism for IL-15-Driven B Cell Chronic Lymphocytic Leukemia Cycling: Roles for AKT and STAT5 in Modulating Cyclin D2 and DNA Damage Response Proteins.J Immunol. 2019 May 15;202(10):2924-2944. doi: 10.4049/jimmunol.1801142. Epub 2019 Apr 15. J Immunol. 2019. PMID: 30988120 Free PMC article.
-
TLR-9 and IL-15 Synergy Promotes the In Vitro Clonal Expansion of Chronic Lymphocytic Leukemia B Cells.J Immunol. 2015 Aug 1;195(3):901-23. doi: 10.4049/jimmunol.1403189. Epub 2015 Jul 1. J Immunol. 2015. PMID: 26136429 Free PMC article.
-
Immunostimulatory CpG-oligonucleotides cause proliferation, cytokine production, and an immunogenic phenotype in chronic lymphocytic leukemia B cells.Blood. 2000 Feb 1;95(3):999-1006. Blood. 2000. PMID: 10648415
-
Effect of immunostimulatory CpG-oligonucleotides in chronic lymphocytic leukemia B cells.Leuk Lymphoma. 2001 Jul;42(3):301-7. doi: 10.3109/10428190109064586. Leuk Lymphoma. 2001. PMID: 11699394 Review.
-
The pathogenesis of chronic lymphocytic leukemia.Annu Rev Pathol. 2014;9:103-18. doi: 10.1146/annurev-pathol-020712-163955. Epub 2013 Aug 26. Annu Rev Pathol. 2014. PMID: 23987584 Free PMC article. Review.
Cited by
-
Dual stimulation by autoantigen and CpG fosters the proliferation of exhausted rheumatoid factor-specific CD21low B cells in hepatitis C virus-cured mixed cryoglobulinemia.Front Immunol. 2023 Feb 8;14:1094871. doi: 10.3389/fimmu.2023.1094871. eCollection 2023. Front Immunol. 2023. PMID: 36845129 Free PMC article.
-
Mechanism for IL-15-Driven B Cell Chronic Lymphocytic Leukemia Cycling: Roles for AKT and STAT5 in Modulating Cyclin D2 and DNA Damage Response Proteins.J Immunol. 2019 May 15;202(10):2924-2944. doi: 10.4049/jimmunol.1801142. Epub 2019 Apr 15. J Immunol. 2019. PMID: 30988120 Free PMC article.
-
Ibrutinib and venetoclax target distinct subpopulations of CLL cells: implication for residual disease eradication.Blood Cancer J. 2021 Feb 18;11(2):39. doi: 10.1038/s41408-021-00429-z. Blood Cancer J. 2021. PMID: 33602908 Free PMC article.
-
The Role of the Microenvironment and Cell Adhesion Molecules in Chronic Lymphocytic Leukemia.Cancers (Basel). 2023 Oct 26;15(21):5160. doi: 10.3390/cancers15215160. Cancers (Basel). 2023. PMID: 37958334 Free PMC article. Review.
-
Chronic inflammation: key player and biomarker-set to predict and prevent cancer development and progression based on individualized patient profiles.EPMA J. 2019 Nov 20;10(4):365-381. doi: 10.1007/s13167-019-00194-x. eCollection 2019 Dec. EPMA J. 2019. PMID: 31832112 Free PMC article. Review.
References
-
- Nabhan C, Raca G, Wang YL. Predicting Prognosis in Chronic Lymphocytic Leukemia in the Contemporary Era. JAMA Oncol. 2015;1:965–974. - PubMed
-
- Duhren-von Minden M, Ubelhart R, Schneider D, Wossning T, Bach MP, Buchner M, Hofmann D, Surova E, Follo M, Kohler F, Wardemann H, Zirlik K, Veelken H, Jumaa H. Chronic lymphocytic leukaemia is driven by antigen-independent cell-autonomous signalling. Nature. 2012;489:309–312. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

