Bridging immunogenetics and immunoproteomics: Model positional scanning library analysis for Major Histocompatibility Complex class II DQ in Tursiops truncatus
- PMID: 30070993
- PMCID: PMC6072028
- DOI: 10.1371/journal.pone.0201299
Bridging immunogenetics and immunoproteomics: Model positional scanning library analysis for Major Histocompatibility Complex class II DQ in Tursiops truncatus
Abstract
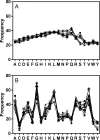
The Major Histocompatibility Complex (MHC) is a critical element in mounting an effective immune response in vertebrates against invading pathogens. Studies of MHC in wildlife populations have typically focused on assessing diversity within the peptide binding regions (PBR) of the MHC class II (MHC II) family, especially the DQ receptor genes. Such metrics of diversity, however, are of limited use to health risk assessment since functional analyses (where changes in the PBR are correlated to recognition/pathologies of known pathogen proteins), are difficult to conduct in wildlife species. Here we describe a means to predict the binding preferences of MHC proteins: We have developed a model positional scanning library analysis (MPSLA) by harnessing the power of mixture based combinatorial libraries to probe the peptide landscapes of distinct MHC II DQ proteins. The algorithm provided by NNAlign was employed to predict the binding affinities of sets of peptides generated for DQ proteins. These binding affinities were then used to retroactively construct a model Positional Scanning Library screen. To test the utility of the approach, a model screen was compared to physical combinatorial screens for human MHC II DP. Model library screens were generated for DQ proteins derived from sequence data from bottlenose dolphins from the Indian River Lagoon (IRL) and the Atlantic coast of Florida, and compared to screens of DQ proteins from Genbank for dolphin and three other cetaceans. To explore the peptide binding landscape for DQ proteins from the IRL, combinations of the amino acids identified as active were compiled into peptide sequence lists that were used to mine databases for representation in known proteins. The frequency of which peptide sequences predicted to bind the MHC protein are found in proteins from pathogens associated with marine mammals was found to be significant (p values <0.0001). Through this analysis, genetic variation in MHC (classes I and II) can now be associated with the binding repertoires of the expressed MHC proteins and subsequently used to identify target pathogens. This approach may be eventually applied to evaluate individual population and species risk for outbreaks of emerging diseases.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures







References
-
- Landry C, Bernatchez L (2001) Comparative analysis of population structure across environments and geographical scales at major histocompatibility complex and microsatellite loci in Atlantic salmon (Salmo salar). Mol Ecol 10: 2525–2539. 1383 [pii]. - PubMed
-
- Bernatchez L, Landry C (2003) MHC studies in nonmodel vertebrates: what have we learned about natural selection in 15 years? J Evol Biol 16: 363–377. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

