Liraglutide treatment improves postprandial lipid metabolism and cardiometabolic risk factors in humans with adequately controlled type 2 diabetes: A single-centre randomized controlled study
- PMID: 30073766
- PMCID: PMC6585708
- DOI: 10.1111/dom.13487
Liraglutide treatment improves postprandial lipid metabolism and cardiometabolic risk factors in humans with adequately controlled type 2 diabetes: A single-centre randomized controlled study
Abstract
Aims: Patients with type 2 diabetes and non-alcoholic fatty liver disease (NAFLD) exhibit considerable residual risk for cardiovascular disease (CVD). There is, therefore, increasing interest in targeting postprandial lipid metabolism and remnant cholesterol. Treatment with the glucagon-like peptide 1 (GLP-1) analogue liraglutide reduces CVD risk by mechanisms that remain unexplained in part. Here we investigated the effects of liraglutide intervention on ectopic fat depots, hepatic lipogenesis and fat oxidation, postprandial lipid metabolism and glycaemia in humans with type 2 diabetes.
Methods: The effect of liraglutide was investigated in 22 patients with adequately controlled type 2 diabetes. Patients were randomly allocated, in a single-blind fashion, to either liraglutide 1.8 mg or placebo once daily for 16 weeks. Because liraglutide is known to promote weight loss, the study included dietary counselling to achieve similar weight loss in the liraglutide and placebo groups. Cardiometabolic responses to a high-fat mixed meal were measured before and at the end of the liraglutide intervention.
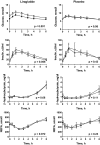
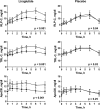
Results: Weight loss at Week 16 was similar between the groups: -2.4 kg (-2.5%) in the liraglutide group and -2.1 kg (-2.2%) in the placebo group. HBA1c improved by 6.4 mmol/mol (0.6%) in the liraglutide group (P = 0.005). Liver fat decreased in both groups, by 31% in the liraglutide group and by 18% in the placebo group, but there were no significant changes in the rate of hepatic de novo lipogenesis or β-hydroxybutyrate levels, a marker of fat oxidation. We observed significant postprandial decreases in triglycerides only in plasma, chylomicrons and VLDL, and remnant particle cholesterol after treatment in the liraglutide group. Fasting and postprandial apoCIII concentrations decreased after liraglutide intervention and these changes were closely related to reduced glycaemia. In relative importance analysis, approximately half of the changes in postprandial lipids were explained by reductions in apoCIII concentrations, whereas less than 10% of the variation in postprandial lipids was explained by reductions in weight, glycaemic control, liver fat or postprandial insulin responses.
Conclusions: Intervention with liraglutide for 16 weeks produces multiple improvements in cardiometabolic risk factors that were not seen in the placebo group, despite similar weight loss. Of particular importance was a marked reduction in postprandial atherogenic remnant particles. The underlying mechanism may be improved glycaemic control, which leads to reduced expression of apoCIII, a key regulator of hypertriglyceridaemia in hyperglycaemic patients.
Keywords: GLP-1-agonist; apolipoprotein C3; atherogenic dyslipidaemia; de novo lipogenesis; liraglutide; liver fat; postprandial lipids; remnant lipoproteins.
© 2018 The Authors. Diabetes, Obesity and Metabolism published by John Wiley & Sons Ltd.
Conflict of interest statement
The authors report no duality of interest.
Figures


References
-
- Wild SH, Walker JJ, Morling JR, et al. Cardiovascular disease, cancer, and mortality among people with type 2 diabetes and alcoholic or nonalcoholic fatty liver disease hospital admission. Diabetes Care. 2018;41:341‐347. - PubMed
-
- Varbo A, Freiberg JJ, Nordestgaard BG. Remnant cholesterol and myocardial infarction in normal weight, overweight, and obese individuals from the Copenhagen General Population Study. Clin Chem. 2018;64:219‐230. - PubMed
-
- Varbo A, Benn M, Nordestgaard BG. Remnant cholesterol as a cause of ischemic heart disease: evidence, definition, measurement, atherogenicity, high risk patients, and present and future treatment. Pharmacol Ther. 2014;141:358‐367. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

