Universal Influenza Vaccines: Progress in Achieving Broad Cross-Protection In Vivo
- PMID: 30084906
- PMCID: PMC6887936
- DOI: 10.1093/aje/kwy145
Universal Influenza Vaccines: Progress in Achieving Broad Cross-Protection In Vivo
Abstract
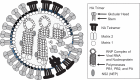
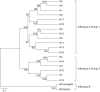
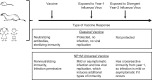
Despite all we have learned since 1918 about influenza virus and immunity, available influenza vaccines remain inadequate to control outbreaks of unexpected strains. Universal vaccines not requiring strain matching would be a major improvement. Their composition would be independent of predicting circulating viruses and thus potentially effective against unexpected drift or pandemic strains. This commentary explores progress with candidate universal vaccines based on various target antigens. Candidates include vaccines based on conserved viral proteins such as nucleoprotein and matrix, on the conserved hemagglutinin (HA) stem, and various combinations. Discussion covers the differing evidence for each candidate vaccine demonstrating protection in animals against influenza viruses of widely divergent HA subtypes and groups; durability of protection; routes of administration, including mucosal, providing local immunity; and reduction of transmission. Human trials of some candidate universal vaccines have been completed or are underway. Interestingly, the HA stem, like nucleoprotein and matrix, induces immunity that permits some virus replication and emergence of escape mutants fit enough to cause disease. Vaccination with multiple target antigens will thus have advantages over use of single antigens. Ultimately, a universal vaccine providing long-term protection against all influenza virus strains might contribute to pandemic control and routine vaccination.
Figures
Similar articles
-
Universal influenza vaccines, science fiction or soon reality?Expert Rev Vaccines. 2015;14(10):1299-301. doi: 10.1586/14760584.2015.1060860. Epub 2015 Jun 23. Expert Rev Vaccines. 2015. PMID: 26104835
-
Neuraminidase-Inhibiting Antibody Titers Correlate with Protection from Heterologous Influenza Virus Strains of the Same Neuraminidase Subtype.J Virol. 2018 Aug 16;92(17):e01006-18. doi: 10.1128/JVI.01006-18. Print 2018 Sep 1. J Virol. 2018. PMID: 29925654 Free PMC article.
-
Virus-like particles as universal influenza vaccines.Expert Rev Vaccines. 2012 Aug;11(8):995-1007. doi: 10.1586/erv.12.70. Expert Rev Vaccines. 2012. PMID: 23002980 Free PMC article. Review.
-
Vaccination with Recombinant Parainfluenza Virus 5 Expressing Neuraminidase Protects against Homologous and Heterologous Influenza Virus Challenge.J Virol. 2017 Nov 14;91(23):e01579-17. doi: 10.1128/JVI.01579-17. Print 2017 Dec 1. J Virol. 2017. PMID: 28931689 Free PMC article.
-
Progress in the Development of Universal Influenza Vaccines.Viruses. 2020 Sep 17;12(9):1033. doi: 10.3390/v12091033. Viruses. 2020. PMID: 32957468 Free PMC article. Review.
Cited by
-
The 1918 Influenza Pandemic: Looking Back, Looking Forward.Am J Epidemiol. 2018 Dec 1;187(12):2493-2497. doi: 10.1093/aje/kwy207. Am J Epidemiol. 2018. PMID: 30346477 Free PMC article.
-
Impact of Individual Viral Gene Segments from Influenza A/H5N8 Virus on the Protective Efficacy of Inactivated Subtype-Specific Influenza Vaccine.Pathogens. 2021 Mar 19;10(3):368. doi: 10.3390/pathogens10030368. Pathogens. 2021. PMID: 33808583 Free PMC article.
-
Beyond clinical trials: Evolutionary and epidemiological considerations for development of a universal influenza vaccine.PLoS Pathog. 2020 Sep 24;16(9):e1008583. doi: 10.1371/journal.ppat.1008583. eCollection 2020 Sep. PLoS Pathog. 2020. PMID: 32970783 Free PMC article. Review.
-
The effect of respiratory viruses on immunogenicity and protection induced by a candidate universal influenza vaccine in mice.PLoS One. 2019 Apr 15;14(4):e0215321. doi: 10.1371/journal.pone.0215321. eCollection 2019. PLoS One. 2019. PMID: 30986224 Free PMC article.
-
Development of a Universal Influenza Vaccine.J Immunol. 2019 Jan 15;202(2):392-398. doi: 10.4049/jimmunol.1801054. J Immunol. 2019. PMID: 30617121 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

