Coherence in carotenoid-to-chlorophyll energy transfer
- PMID: 30089871
- PMCID: PMC6082895
- DOI: 10.1038/s41467-018-05596-5
Coherence in carotenoid-to-chlorophyll energy transfer
Abstract
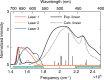
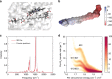
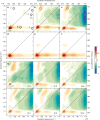
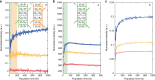
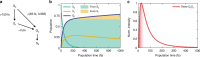
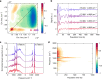
The subtle details of the mechanism of energy flow from carotenoids to chlorophylls in biological light-harvesting complexes are still not fully understood, especially in the ultrafast regime. Here we focus on the antenna complex peridinin-chlorophyll a-protein (PCP), known for its remarkable efficiency of excitation energy transfer from carotenoids-peridinins-to chlorophylls. PCP solutions are studied by means of 2D electronic spectroscopy in different experimental conditions. Together with a global kinetic analysis and multiscale quantum chemical calculations, these data allow us to comprehensively address the contribution of the potential pathways of energy flow in PCP. These data support dominant energy transfer from peridinin S2 to chlorophyll Qy state via an ultrafast coherent mechanism. The coherent superposition of the two states is functional to drive population to the final acceptor state, adding an important piece of information in the quest for connections between coherent phenomena and biological functions.
Conflict of interest statement
The authors declare no competing interests.
Figures
Similar articles
-
Structural and spectroscopic characterization of the peridinin-chlorophyll a-protein (PCP) complex from Heterocapsa pygmaea (HPPCP).Biochim Biophys Acta Bioenerg. 2025 Jan 1;1866(1):149510. doi: 10.1016/j.bbabio.2024.149510. Epub 2024 Sep 24. Biochim Biophys Acta Bioenerg. 2025. PMID: 39321862
-
Excitation transfer in the peridinin-chlorophyll-protein of Amphidinium carterae.Biophys J. 2000 Oct;79(4):1695-705. doi: 10.1016/S0006-3495(00)76422-8. Biophys J. 2000. PMID: 11023878 Free PMC article.
-
Molecular factors controlling photosynthetic light harvesting by carotenoids.Acc Chem Res. 2010 Aug 17;43(8):1125-34. doi: 10.1021/ar100030m. Acc Chem Res. 2010. PMID: 20446691 Free PMC article.
-
Spectroscopy of the peridinin-chlorophyll-a protein: insight into light-harvesting strategy of marine algae.Arch Biochem Biophys. 2007 Feb 15;458(2):111-20. doi: 10.1016/j.abb.2006.10.006. Epub 2006 Oct 27. Arch Biochem Biophys. 2007. PMID: 17098207 Review.
-
The unique photophysical properties of the Peridinin-Chlorophyll-α-Protein.Curr Protein Pept Sci. 2014;15(4):332-50. doi: 10.2174/1389203715666140327111139. Curr Protein Pept Sci. 2014. PMID: 24678668 Free PMC article. Review.
Cited by
-
Photocurrent-Detected 2D Electronic Spectroscopy Reveals Ultrafast Hole Transfer in Operating PM6/Y6 Organic Solar Cells.J Phys Chem Lett. 2021 Apr 29;12(16):3983-3988. doi: 10.1021/acs.jpclett.1c00822. Epub 2021 Apr 20. J Phys Chem Lett. 2021. PMID: 33877838 Free PMC article.
-
Light-Harvesting: The Never-Ending Lesson of Nature.ACS Cent Sci. 2022 Mar 23;8(3):306-308. doi: 10.1021/acscentsci.2c00137. Epub 2022 Feb 21. ACS Cent Sci. 2022. PMID: 35350603 Free PMC article. No abstract available.
-
Carotenoid-Chlorophyll Interactions in a Photosynthetic Antenna Protein: A Supramolecular QM/MM Approach.Molecules. 2018 Oct 10;23(10):2589. doi: 10.3390/molecules23102589. Molecules. 2018. PMID: 30308965 Free PMC article.
-
Quantum Chemical Modeling of the Photoinduced Activity of Multichromophoric Biosystems.Chem Rev. 2019 Aug 28;119(16):9361-9380. doi: 10.1021/acs.chemrev.9b00135. Epub 2019 Jul 5. Chem Rev. 2019. PMID: 31276384 Free PMC article. Review.
-
In Silico Ultrafast Nonlinear Spectroscopy Meets Experiments: The Case of Perylene Bisimide Dye.J Chem Theory Comput. 2021 Nov 9;17(11):7134-7145. doi: 10.1021/acs.jctc.1c00570. Epub 2021 Oct 22. J Chem Theory Comput. 2021. PMID: 34676761 Free PMC article.
References
-
- Bautista JA, et al. Singlet and triplet energy transfer in the peridinin-chlorophyll a-protein from Amphidinium carterae. J. Phys. Chem. A. 1999;103:2267–2273. doi: 10.1021/jp983943f. - DOI
-
- van Stokkum IHM, et al. Inter-pigment interactions in the peridinin chlorophyll protein studied by global and target analysis of time resolved absorption spectra. Chem. Phys. 2009;357:70–78. doi: 10.1016/j.chemphys.2008.10.005. - DOI
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

