Infectious Complications of Multiple Sclerosis Therapies: Implications for Screening, Prophylaxis, and Management
- PMID: 30094293
- PMCID: PMC6080056
- DOI: 10.1093/ofid/ofy174
Infectious Complications of Multiple Sclerosis Therapies: Implications for Screening, Prophylaxis, and Management
Abstract
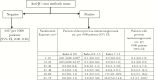
Multiple sclerosis therapies include interferons, glatiramer, and multiple immunosuppressive drugs. Discerning infectious risks of immunosuppressive drugs requires understanding their mechanisms of action and analyzing interventional studies and postmarketing observational data. Though identical immunosuppressive therapies are sometimes used in non-neurologic conditions, infectious risks may differ in this population. Screening for and treatment of latent tuberculosis (TB) infection should be prioritized for patients receiving alemtuzumab; ocrelizumab is likely not associated with an increased risk of TB. Hepatitis B virus (HBV) reactivation can be devastating for patients treated with ocrelizumab and alemtuzumab, whereas the small molecule oral agents do not likely pose substantial risk of HBV. Progressive multifocal leukoencephalopathy is a particular concern with natalizumab. Alemtuzumab, and possibly natalizumab and fingolimod, risks herpes virus reactivation and may warrant prophylaxis. Unusual opportunistic infections have been described. Vaccination is an important tool in preventing infections, though vaccine timing and contraindications can be complex.
Keywords: immunosuppression; multiple sclerosis; opportunistic infections.
Figures
Comment in
-
Corrigendum.Open Forum Infect Dis. 2018 Nov 26;5(11):ofy279. doi: 10.1093/ofid/ofy279. eCollection 2018 Nov. Open Forum Infect Dis. 2018. PMID: 30539035 Free PMC article.
References
-
- Brooks M. MS drug Daclizumab (zinbryta) pulled from the market. Medscape Medical News; 2 March 2018. Available at: http://www.medscape.com/viewarticle/893352. Accessed 6 March 2018.
-
- Kappos L, Bar-Or A, Cree BAC, et al. ; EXPAND Clinical Investigators Siponimod versus placebo in secondary progressive multiple sclerosis (EXPAND): a double-blind, randomised, phase 3 study. Lancet 2018; 391:1263–73. - PubMed
-
- Rudick RA, Stuart WH, Calabresi PA, et al. ; SENTINEL Investigators. Natalizumab plus interferon beta-1a for relapsing multiple sclerosis N Engl J Med 2006; 354:911–23. - PubMed
-
- Hawker K, O’Connor P, Freedman MS, et al. ; OLYMPUS trial group Rituximab in patients with primary progressive multiple sclerosis: results of a randomized double-blind placebo-controlled multicenter trial. Ann Neurol 2009; 66:460–71. - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

