Generation of Tumor-Reactive T Cells by Co-culture of Peripheral Blood Lymphocytes and Tumor Organoids
- PMID: 30100188
- PMCID: PMC6558289
- DOI: 10.1016/j.cell.2018.07.009
Generation of Tumor-Reactive T Cells by Co-culture of Peripheral Blood Lymphocytes and Tumor Organoids
Abstract
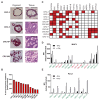
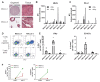
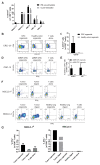
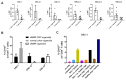
Cancer immunotherapies have shown substantial clinical activity for a subset of patients with epithelial cancers. Still, technological platforms to study cancer T-cell interactions for individual patients and understand determinants of responsiveness are presently lacking. Here, we establish and validate a platform to induce and analyze tumor-specific T cell responses to epithelial cancers in a personalized manner. We demonstrate that co-cultures of autologous tumor organoids and peripheral blood lymphocytes can be used to enrich tumor-reactive T cells from peripheral blood of patients with mismatch repair-deficient colorectal cancer and non-small-cell lung cancer. Furthermore, we demonstrate that these T cells can be used to assess the efficiency of killing of matched tumor organoids. This platform provides an unbiased strategy for the isolation of tumor-reactive T cells and provides a means by which to assess the sensitivity of tumor cells to T cell-mediated attack at the level of the individual patient.
Keywords: T cell; adoptive cell transfer; colorectal cancer; immune checkpoint blockade; immunotherapy; microsatellite instable; mismatch repair deficient; non-small cell lung cancer; organoids.
Copyright © 2018 Elsevier Inc. All rights reserved.
Conflict of interest statement
H.C. is inventor on several patents related to organoid technology.
N.S. reports grants from The Netherlands Organisation for Scientific Research, during the conduct of the study; other from Vertex Pharmaceuticals Incorporated, outside the submitted work; In addition, N.S. has a patent PCT/EP2015/077990 with royalties paid to Stichting HUB, and a patent PCT/EP2015/077988 with royalties paid to Stichting HUB.
Figures

Comment in
-
Studying tumour-specific T cell responses in 3D.Nat Rev Immunol. 2018 Oct;18(10):602-603. doi: 10.1038/s41577-018-0058-7. Nat Rev Immunol. 2018. PMID: 30116005 No abstract available.
-
Studying Tumor-ReacTive T Cells: A Personalized Organoid Model.Cell Stem Cell. 2018 Sep 6;23(3):318-319. doi: 10.1016/j.stem.2018.08.015. Cell Stem Cell. 2018. PMID: 30193129
References
-
- Blank CU, Haanen JB, Ribas A, Schumacher TN. CANCER IMMUNOLOGY. The “cancer immunogram”. Science. 2016;352:658–660. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

