Recombinant Antibodies in Veterinary Medicine: An Update
- PMID: 30101148
- PMCID: PMC6072837
- DOI: 10.3389/fvets.2018.00175
Recombinant Antibodies in Veterinary Medicine: An Update
Abstract
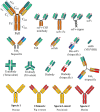
The production of recombinant antibodies has had a tremendous impact on several research fields, most prominently in biotechnology, immunology and medicine, enabling enormous advances in each. Thus far, a broad diversity of recombinant antibody (rAb) forms have been designed and expressed using different expression systems. Even though the majority of rAbs approved for clinical use are targeted to humans, advances in veterinary medicine seem promising. The aim of this mini-review is to present an update regarding the rAbs in veterinary medicine reported to date, as well as their potential use in diagnostics, prophylaxis and therapeutics. Full- and single-chain fragment variables are the most common forms of rAbs developed for the detection, prevention and control of parasitic, bacterial and viral diseases, as well as pain and cancer treatment. Nonetheless, advances in research seem to be skewed toward economically important animals, such as pigs, cows, poultry and dogs. Although significant results have been obtained from the rAbs reported here, most have not been developed enough to be approved. Further research and clinical trials should be encouraged to enable important findings to fulfill their intended potential to improve animal well-being.
Keywords: biotechnology; chimeric antibodies; nanobodies; recombinant antibodies; single-chain antibodies; veterinary medicine.
Figures
Similar articles
-
Recombinant protein-based viral disease diagnostics in veterinary medicine.Expert Rev Mol Diagn. 2010 Sep;10(6):731-53. doi: 10.1586/erm.10.61. Expert Rev Mol Diagn. 2010. PMID: 20843198 Review.
-
Applications of single-chain variable fragment antibodies in therapeutics and diagnostics.Biotechnol Adv. 2009 Jul-Aug;27(4):502-20. doi: 10.1016/j.biotechadv.2009.04.004. Epub 2009 Apr 15. Biotechnol Adv. 2009. PMID: 19374944 Review.
-
Theranostics Aspects of Various Nanoparticles in Veterinary Medicine.Int J Mol Sci. 2018 Oct 24;19(11):3299. doi: 10.3390/ijms19113299. Int J Mol Sci. 2018. PMID: 30352960 Free PMC article. Review.
-
Recent progress in biomolecular engineering.Biotechnol Prog. 2000 Jan-Feb;16(1):2-16. doi: 10.1021/bp088059d. Biotechnol Prog. 2000. PMID: 10662483 Review.
-
Recombinant Antibody Fragments for Neurodegenerative Diseases.Curr Neuropharmacol. 2017;15(5):779-788. doi: 10.2174/1570159X01666160930121647. Curr Neuropharmacol. 2017. PMID: 27697033 Free PMC article. Review.
Cited by
-
Construction and characterization of porcine single-chain fragment variable antibodies that neutralize transmissible gastroenteritis virus in vitro.Arch Virol. 2019 Apr;164(4):983-994. doi: 10.1007/s00705-019-04156-6. Epub 2019 Feb 7. Arch Virol. 2019. PMID: 30729994 Free PMC article.
-
Expansion of platform physiologically-based pharmacokinetic model for monoclonal antibodies towards different preclinical species: cats, sheep, and dogs.J Pharmacokinet Pharmacodyn. 2024 Dec;51(6):621-638. doi: 10.1007/s10928-023-09893-5. Epub 2023 Nov 10. J Pharmacokinet Pharmacodyn. 2024. PMID: 37947924
-
Evaluation of a Recombinant Mouse X Pig Chimeric Anti-Porcine DEC205 Antibody Fused with Structural and Nonstructural Peptides of PRRS Virus.Vaccines (Basel). 2019 May 23;7(2):43. doi: 10.3390/vaccines7020043. Vaccines (Basel). 2019. PMID: 31126125 Free PMC article.
-
A novel antibody treatment reduces deformed wing virus loads in the western honey bee (Apis mellifera).mSphere. 2024 Nov 21;9(11):e0049724. doi: 10.1128/msphere.00497-24. Epub 2024 Oct 30. mSphere. 2024. PMID: 39475320 Free PMC article.
-
Review on Preventive Measures to Reduce Post-Weaning Diarrhoea in Piglets.Animals (Basel). 2022 Sep 27;12(19):2585. doi: 10.3390/ani12192585. Animals (Basel). 2022. PMID: 36230326 Free PMC article. Review.
References
-
- Shoemaker CB. When Will rAbs Replace mAbs in Labs? WB Saunders; (2005). - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources

