Colorimetric Carbonyl Sulfide (COS)/Hydrogen Sulfide (H2 S) Donation from γ-Ketothiocarbamate Donor Motifs
- PMID: 30102448
- PMCID: PMC6504913
- DOI: 10.1002/anie.201806854
Colorimetric Carbonyl Sulfide (COS)/Hydrogen Sulfide (H2 S) Donation from γ-Ketothiocarbamate Donor Motifs
Abstract
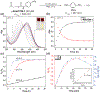
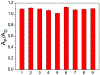
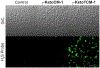
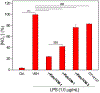
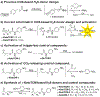
Hydrogen sulfide (H2 S) is a biologically active molecule that exhibits protective effects in a variety of physiological and pathological processes. Although several H2 S-related biological effects have been discovered by using H2 S donors, knowing how much H2 S has been released from donors under different conditions remains challenging. Now, a series of γ-ketothiocarbamate (γ-KetoTCM) compounds that provide the first examples of colorimetric H2 S donors and enable direct quantification of H2 S release, were reported. These compounds are activated through a pH-dependent deprotonation/β-elimination sequence to release carbonyl sulfide (COS), which is quickly converted into H2 S by carbonic anhydrase. The p-nitroaniline released upon donor activation provides an optical readout that correlates directly to COS/H2 S release, thus enabling colorimetric measurement of H2 S donation.
Keywords: anti-inflammation; carbonyl sulfide; colorimetry; hydrogen sulfide; γ-ketothiocarbamate.
© 2018 Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim.
Figures

References
-
- Pluth MD, Bailey TS, Hammers MD, Hartle MD, Henthorn HA, Steiger AK, Synlett 2015, 26, 2633–2643;
- Zhao Y, Biggs TD, Xian M, Chem. Commun. 2014, 50, 11788–11805; - PMC - PubMed
- Zhao Y, Pacheco A, Xian M, Handb. Exp. Pharmacol. 2015, 230, 365–388; - PubMed
- Zheng Y, Yu B, De La Cruz LK, Roy Choudhury M, Anifowose A, Wang B, Med. Res. Rev. 2018, 38, 57–100; - PubMed
- Szabo C, Papapetropoulos A, Pharmacol. Rev. 2017, 69, 497–564. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

