Combinations of DIPs and Dprs control organization of olfactory receptor neuron terminals in Drosophila
- PMID: 30102700
- PMCID: PMC6107282
- DOI: 10.1371/journal.pgen.1007560
Combinations of DIPs and Dprs control organization of olfactory receptor neuron terminals in Drosophila
Abstract
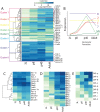
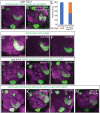
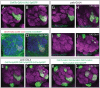
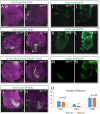
In Drosophila, 50 classes of olfactory receptor neurons (ORNs) connect to 50 class-specific and uniquely positioned glomeruli in the antennal lobe. Despite the identification of cell surface receptors regulating axon guidance, how ORN axons sort to form 50 stereotypical glomeruli remains unclear. Here we show that the heterophilic cell adhesion proteins, DIPs and Dprs, are expressed in ORNs during glomerular formation. Many ORN classes express a unique combination of DIPs/dprs, with neurons of the same class expressing interacting partners, suggesting a role in class-specific self-adhesion between ORN axons. Analysis of DIP/Dpr expression revealed that ORNs that target neighboring glomeruli have different combinations, and ORNs with very similar DIP/Dpr combinations can project to distant glomeruli in the antennal lobe. DIP/Dpr profiles are dynamic during development and correlate with sensilla type lineage for some ORN classes. Perturbations of DIP/dpr gene function result in local projection defects of ORN axons and glomerular positioning, without altering correct matching of ORNs with their target neurons. Our results suggest that context-dependent differential adhesion through DIP/Dpr combinations regulate self-adhesion and sort ORN axons into uniquely positioned glomeruli.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures



References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

