Cationic High Molecular Weight Lignin Polymer: A Flocculant for the Removal of Anionic Azo-Dyes from Simulated Wastewater
- PMID: 30103485
- PMCID: PMC6222342
- DOI: 10.3390/molecules23082005
Cationic High Molecular Weight Lignin Polymer: A Flocculant for the Removal of Anionic Azo-Dyes from Simulated Wastewater
Abstract
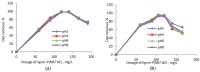
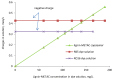
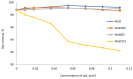
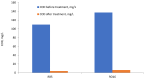
The presence of dyes in wastewater effluents made from the textile industry is a major environmental problem due to their complex structure and poor biodegradability. In this study, a cationic lignin polymer was synthesized via the free radical polymerization of lignin with [2-(methacryloyloxy) ethyl] trimethyl ammonium chloride (METAC) and used to remove anionic azo-dyes (reactive black 5, RB5, and reactive orange 16, RO16) from simulated wastewater. The effects of pH, salt, and concentration of dyes, as well as the charge density and molecular weight of lignin-METAC polymer on dye removal were examined. Results demonstrated that lignin-METAC was an effective flocculant for the removal of dye via charge neutralization and bridging mechanisms. The dye removal efficiency of lignin-METAC polymer was independent of pH. The dosage of the lignin polymer required for reaching the maximum removal had a linear relationship with the dye concentration. The presence of inorganic salts including NaCl, NaNO₃, and Na₂SO₄ had a marginal effect on the dye removal. Under the optimized conditions, greater than 98% of RB5 and 94% of RO16 were removed at lignin-METAC concentrations of 120 mg/L and 105 mg/L in the dye solutions, respectively.
Keywords: COD; azo dye; flocculation; lignin modification; lignin-METAC.
Conflict of interest statement
The authors declare no conflict of interest.
Figures






References
-
- Rasool K., Woo S.H., Lee D.S. Simultaneous removal of COD and Direct Red 80 in a mixed anaerobic sulfate-reducing bacteria culture. Chem. Eng. J. 2013;223:611–616. doi: 10.1016/j.cej.2013.03.031. - DOI
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

