Hydrogen Sulfide-Releasing Fibrous Membranes: Potential Patches for Stimulating Human Stem Cells Proliferation and Viability under Oxidative Stress
- PMID: 30103516
- PMCID: PMC6121677
- DOI: 10.3390/ijms19082368
Hydrogen Sulfide-Releasing Fibrous Membranes: Potential Patches for Stimulating Human Stem Cells Proliferation and Viability under Oxidative Stress
Abstract
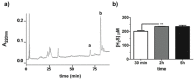
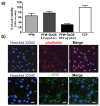
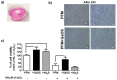
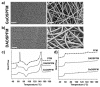
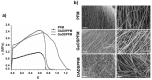
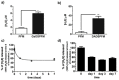
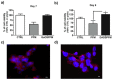
The design of biomaterial platforms able to release bioactive molecules is mandatory in tissue repair and regenerative medicine. In this context, electrospinning is a user-friendly, versatile and low-cost technique, able to process different kinds of materials in micro- and nano-fibers with a large surface area-to-volume ratio for an optimal release of gaseous signaling molecules. Recently, the antioxidant and anti-inflammatory properties of the endogenous gasotramsmitter hydrogen sulfide (H₂S), as well as its ability to stimulate relevant biochemical processes on the growth of mesenchymal stem cells (MSC), have been investigated. Therefore, in this work, new poly(lactic) acid fibrous membranes (PFM), doped and functionalized with H₂S slow-releasing donors extracted from garlic, were synthetized. These innovative H₂S-releasing mats were characterized for their morphological, thermal, mechanical, and biological properties. Their antimicrobial activity and effects on the in vitro human cardiac MSC growth, either in the presence or in the absence of oxidative stress, were here assessed. On the basis of the results here presented, these new H₂S-releasing PFM could represent promising and low-cost scaffolds or patches for biomedical applications in tissue repair.
Keywords: PLA fibers; antibacterial properties; cytotoxicity; garlic extracts; mesenchymal stem cells; microstructure; organosulfur compounds; thermal and mechanical properties.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


Similar articles
-
Glutathione-Allylsulfur Conjugates as Mesenchymal Stem Cells Stimulating Agents for Potential Applications in Tissue Repair.Int J Mol Sci. 2020 Feb 28;21(5):1638. doi: 10.3390/ijms21051638. Int J Mol Sci. 2020. PMID: 32121252 Free PMC article.
-
Glutathione-garlic sulfur conjugates: slow hydrogen sulfide releasing agents for therapeutic applications.Molecules. 2015 Jan 20;20(1):1731-50. doi: 10.3390/molecules20011731. Molecules. 2015. PMID: 25608858 Free PMC article.
-
Biological thiols-triggered hydrogen sulfide releasing microfibers for tissue engineering applications.Acta Biomater. 2015 Nov;27:205-213. doi: 10.1016/j.actbio.2015.09.010. Epub 2015 Sep 9. Acta Biomater. 2015. PMID: 26363376 Free PMC article.
-
Immunomodulatory Effects of Glutathione, Garlic Derivatives, and Hydrogen Sulfide.Nutrients. 2019 Jan 30;11(2):295. doi: 10.3390/nu11020295. Nutrients. 2019. PMID: 30704060 Free PMC article. Review.
-
Garlic-derived natural polysulfanes as hydrogen sulfide donors: Friend or foe?Food Chem Toxicol. 2016 Sep;95:219-33. doi: 10.1016/j.fct.2016.07.016. Epub 2016 Jul 16. Food Chem Toxicol. 2016. PMID: 27430419 Review.
Cited by
-
Targeting the redox system for cardiovascular regeneration in aging.Aging Cell. 2023 Dec;22(12):e14020. doi: 10.1111/acel.14020. Epub 2023 Nov 13. Aging Cell. 2023. PMID: 37957823 Free PMC article. Review.
-
A biomimetic orthogonal-bilayer tubular scaffold for the co-culture of endothelial cells and smooth muscle cells.RSC Adv. 2021 Sep 27;11(50):31783-31790. doi: 10.1039/d1ra04472a. eCollection 2021 Sep 21. RSC Adv. 2021. PMID: 35496878 Free PMC article.
-
Bacteria for Bioplastics: Progress, Applications, and Challenges.ACS Omega. 2024 Feb 12;9(8):8666-8686. doi: 10.1021/acsomega.3c07372. eCollection 2024 Feb 27. ACS Omega. 2024. PMID: 38434856 Free PMC article. Review.
-
Controlled Release of H2S from Biomimetic Silk Fibroin-PLGA Multilayer Electrospun Scaffolds.Biomacromolecules. 2023 Mar 13;24(3):1366-1376. doi: 10.1021/acs.biomac.2c01383. Epub 2023 Feb 7. Biomacromolecules. 2023. PMID: 36749903 Free PMC article.
-
Functional electrospun nanofibers: fabrication, properties, and applications in wound-healing process.RSC Adv. 2024 Jan 22;14(5):3359-3378. doi: 10.1039/d3ra07075a. eCollection 2024 Jan 17. RSC Adv. 2024. PMID: 38259986 Free PMC article. Review.
References
-
- Mitsuhashi H., Yamashita S., Ikeuchi H., Kuroiwa T., Kaneko Y., Hiromura K., Ueki K., Nojima Y. Oxidative stress-dependent conversion of hydrogen sulfide to sulfite by activated neutrophils. Shock. 2005;24:529–534. doi: 10.1097/01.shk.0000183393.83272.de. - DOI - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

