EGFR may participate in immune evasion through regulation of B7‑H5 expression in non‑small cell lung carcinoma
- PMID: 30106102
- PMCID: PMC6131583
- DOI: 10.3892/mmr.2018.9361
EGFR may participate in immune evasion through regulation of B7‑H5 expression in non‑small cell lung carcinoma
Abstract
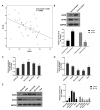
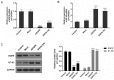
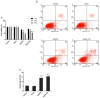
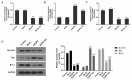
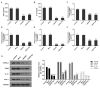
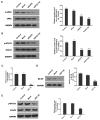
Lung cancer is one of the most prevalent malignancies worldwide; it has been ranked the most lethal type of cancer. Non‑small cell lung carcinoma (NSCLC) comprises >80% of all types of lung cancer. Although certain achievements have been made in the treatment of NSCLC, including the targeted gene drug epidermal growth factor receptor‑tyrosine kinase inhibitor (EGFR‑TKI), the five‑year survival rate of patients has not significantly increased. A previous study demonstrated that B7‑H5, a novel co‑stimulatory molecule in the B7 molecule family, was negatively correlated with EGFR in pancreatic cancer. Thus, in the present study, we aimed to investigate whether EGFR participates in immune evasion, probably through regulation of B7‑H5 expression. NCI‑H1299 NSCLCL cells were divided into control, mock, small interfering‑EGFR and EGFR‑TKI groups. The cell viability and apoptosis rate were analysed by a Cell Counting Kit‑8 assay and flow cytometry. The transforming growth factor (TGF)‑β and interleukin (IL)‑10 content was measured using an ELISA. The expression levels of EGFR, B7‑H5, Survivin, apoptosis regulator Bax, apoptosis regulator Bcl‑2 (Bcl‑2), TGF‑β, vascular endothelial growth factor (VEGF), IL‑10 and cyclooxygenase (COX)‑2 were assessed via quantitative PCR and western blotting. The activation of the tyrosine‑protein kinase JAK2 (JAK2)/signal transducer and activator of transcription 3 (STAT3) signalling pathway was detected using western blotting. The results demonstrated a notable negative correlation between EGFR and B7‑H5 expression levels in cancer tissues and cell lines. Inhibition of EGFR expression via gene silencing and EGFR inhibition markedly decreased cell viability and increased the apoptosis of NCI‑H1299 cells, by upregulating survivin and Bcl‑2 expression. The protein expression levels of TGF‑β, VEGF, IL‑10 and COX‑2 were additionally decreased, with weak activation of the JAK2/STAT3 signalling pathway. EGFR may be involved in immune evasion, possibly through regulation of B7‑H5 expression in NSCLC.
Figures
Similar articles
-
Cyclooxygenase-2-dependent activation of signal transducer and activator of transcription 3 by interleukin-6 in non-small cell lung cancer.Clin Cancer Res. 2005 Nov 1;11(21):7674-82. doi: 10.1158/1078-0432.CCR-05-1205. Clin Cancer Res. 2005. PMID: 16278387
-
Upregulation of PD-L1 by EGFR Activation Mediates the Immune Escape in EGFR-Driven NSCLC: Implication for Optional Immune Targeted Therapy for NSCLC Patients with EGFR Mutation.J Thorac Oncol. 2015 Jun;10(6):910-23. doi: 10.1097/JTO.0000000000000500. J Thorac Oncol. 2015. PMID: 25658629
-
Overexpression of B7-H3 augments anti-apoptosis of colorectal cancer cells by Jak2-STAT3.World J Gastroenterol. 2015 Feb 14;21(6):1804-13. doi: 10.3748/wjg.v21.i6.1804. World J Gastroenterol. 2015. PMID: 25684945 Free PMC article.
-
Immune checkpoint inhibitors in epidermal growth factor receptor mutant non-small cell lung cancer: Current controversies and future directions.Lung Cancer. 2018 Jan;115:12-20. doi: 10.1016/j.lungcan.2017.11.009. Epub 2017 Nov 13. Lung Cancer. 2018. PMID: 29290252 Review.
-
STAT3: Versatile Functions in Non-Small Cell Lung Cancer.Cancers (Basel). 2020 Apr 29;12(5):1107. doi: 10.3390/cancers12051107. Cancers (Basel). 2020. PMID: 32365499 Free PMC article. Review.
Cited by
-
HHLA2 promotes tumor progression by long non‑coding RNA H19 in human gallbladder cancer.Int J Oncol. 2022 Sep;61(3):112. doi: 10.3892/ijo.2022.5402. Epub 2022 Aug 3. Int J Oncol. 2022. PMID: 35920182 Free PMC article.
-
Comparative Investigation of Composition, Antifungal, and Anti-Inflammatory Effects of the Essential Oil from Three Industrial Hemp Varieties from Italian Cultivation.Antibiotics (Basel). 2021 Mar 22;10(3):334. doi: 10.3390/antibiotics10030334. Antibiotics (Basel). 2021. PMID: 33809983 Free PMC article.
-
Comprehensive characterization of B7 family members in NSCLC and identification of its regulatory network.Sci Rep. 2023 Mar 15;13(1):4311. doi: 10.1038/s41598-022-26776-w. Sci Rep. 2023. PMID: 36922519 Free PMC article.
-
Dried Blood Specimens as an Alternative Specimen for Immune Response Monitoring During HIV Infection: A Proof of Concept and Simple Method in a Pediatric Cohort.Front Med (Lausanne). 2021 Jun 15;8:678850. doi: 10.3389/fmed.2021.678850. eCollection 2021. Front Med (Lausanne). 2021. PMID: 34211989 Free PMC article.
-
KIR3DL3-HHLA2 and TMIGD2-HHLA2 pathways: The dual role of HHLA2 in immune responses and its potential therapeutic approach for cancer immunotherapy.J Adv Res. 2023 May;47:137-150. doi: 10.1016/j.jare.2022.07.013. Epub 2022 Aug 4. J Adv Res. 2023. PMID: 35933091 Free PMC article. Review.
References
-
- Hirsch FR, Varella-Garcia M, Bunn PA, Jr, Di Maria MV, Veve R, Bremmes RM, Baron AE, Zeng C, Franklin WA. Epidermal growth factor receptor in non-small-cell lung carcinomas: Correlation between gene copy number and protein expression and impact on prognosis. J Clin Oncol. 2003;21:3798–3807. doi: 10.1200/JCO.2003.11.069. - DOI - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

