Structure of the mammalian TRPM7, a magnesium channel required during embryonic development
- PMID: 30108148
- PMCID: PMC6126765
- DOI: 10.1073/pnas.1810719115
Structure of the mammalian TRPM7, a magnesium channel required during embryonic development
Abstract
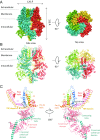
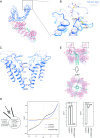
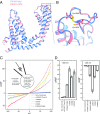
The transient receptor potential ion channel subfamily M, member 7 (TRPM7), is a ubiquitously expressed protein that is required for mouse embryonic development. TRPM7 contains both an ion channel and an α-kinase. The channel domain comprises a nonselective cation channel with notable permeability to Mg2+ and Zn2+ Here, we report the closed state structures of the mouse TRPM7 channel domain in three different ionic conditions to overall resolutions of 3.3, 3.7, and 4.1 Å. The structures reveal key residues for an ion binding site in the selectivity filter, with proposed partially hydrated Mg2+ ions occupying the center of the conduction pore. In high [Mg2+], a prominent external disulfide bond is found in the pore helix, which is essential for ion channel function. Our results provide a structural framework for understanding the TRPM1/3/6/7 subfamily and extend the knowledge base upon which to study the diversity and evolution of TRP channels.
Keywords: TRP channel; chanzyme; ion channel; kinase; zinc.
Copyright © 2018 the Author(s). Published by PNAS.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



Comment in
-
TRPM7 reflected in Cryo-EMirror.Cell Calcium. 2018 Dec;76:129-131. doi: 10.1016/j.ceca.2018.11.004. Epub 2018 Nov 15. Cell Calcium. 2018. PMID: 30470536
References
Publication types
MeSH terms
Substances
Associated data
- Actions
- Actions
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

