HGF-induced formation of the MET-AXL-ELMO2-DOCK180 complex promotes RAC1 activation, receptor clustering, and cancer cell migration and invasion
- PMID: 30108175
- PMCID: PMC6177597
- DOI: 10.1074/jbc.RA118.003063
HGF-induced formation of the MET-AXL-ELMO2-DOCK180 complex promotes RAC1 activation, receptor clustering, and cancer cell migration and invasion
Abstract
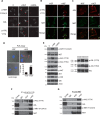
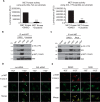
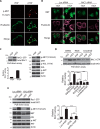
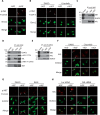
The MET proto-oncogene-encoded receptor tyrosine kinase (MET) and AXL receptor tyrosine kinase (AXL) are independently operating receptor tyrosine kinases (RTKs) that are functionally associated with aggressive and invasive cancer cell growth. However, how MET and AXL regulate the migratory properties of cancer cells remains largely unclear. We report here that the addition of hepatocyte growth factor (HGF), the natural ligand of MET, to serum-starved human glioblastoma cells induces the rapid activation of both MET and AXL and formation of highly polarized MET-AXL clusters on the plasma membrane. HGF also promoted the formation of the MET and AXL protein complexes and phosphorylation of AXL, independent of AXL's ligand, growth arrest-specific 6 (GAS6). The HGF-induced MET-AXL complex stimulated rapid and dynamic cytoskeleton reorganization by activating the small GTPase RAC1, a process requiring both MET and AXL kinase activities. We further found that HGF also promotes the recruitment of ELMO2 and DOCK180, a bipartite guanine nucleotide exchange factor for RAC1, to the MET-AXL complex and thereby stimulates the RAC1-dependent cytoskeleton reorganization. We also demonstrated that the MET-AXL-ELMO2-DOCK180 complex is critical for HGF-induced cell migration and invasion in glioblastoma or other cancer cells. Our findings uncover a critical HGF-dependent signaling pathway that involves the assembly of a large protein complex consisting of MET, AXL, ELMO2, and DOCK180 on the plasma membrane, leading to RAC1-dependent cell migration and invasion in various cancer cells.
Keywords: Ras-related C3 botulinum toxin substrate 1 (Rac1); cancer; cell invasion; cell migration; cell signaling; glioblastoma; guanine nucleotide exchange factor (GEF); receptor tyrosine kinase.
© 2018 Li et al.
Conflict of interest statement
The authors declare that they have no conflicts of interest with the contents of this article
Figures





Similar articles
-
Axl phosphorylates Elmo scaffold proteins to promote Rac activation and cell invasion.Mol Cell Biol. 2015 Jan;35(1):76-87. doi: 10.1128/MCB.00764-14. Epub 2014 Oct 20. Mol Cell Biol. 2015. PMID: 25332238 Free PMC article.
-
AXL and MET crosstalk to promote gonadotropin releasing hormone (GnRH) neuronal cell migration and survival.Mol Cell Endocrinol. 2013 Jul 15;374(1-2):92-100. doi: 10.1016/j.mce.2013.04.018. Epub 2013 May 3. Mol Cell Endocrinol. 2013. PMID: 23648337 Free PMC article.
-
FARP1, ARHGEF39, and TIAM2 are essential receptor tyrosine kinase effectors for Rac1-dependent cell motility in human lung adenocarcinoma.Cell Rep. 2021 Nov 2;37(5):109905. doi: 10.1016/j.celrep.2021.109905. Cell Rep. 2021. PMID: 34731623 Free PMC article.
-
Hepatocyte growth factor/MET in cancer progression and biomarker discovery.Cancer Sci. 2017 Mar;108(3):296-307. doi: 10.1111/cas.13156. Cancer Sci. 2017. PMID: 28064454 Free PMC article. Review.
-
AXL receptor tyrosine kinase as a promising anti-cancer approach: functions, molecular mechanisms and clinical applications.Mol Cancer. 2019 Nov 4;18(1):153. doi: 10.1186/s12943-019-1090-3. Mol Cancer. 2019. PMID: 31684958 Free PMC article. Review.
Cited by
-
Cabozantinib inhibits AXL- and MET-dependent cancer cell migration induced by growth-arrest-specific 6 and hepatocyte growth factor.Biochem Biophys Rep. 2020 Jan 17;21:100726. doi: 10.1016/j.bbrep.2020.100726. eCollection 2020 Mar. Biochem Biophys Rep. 2020. PMID: 32055714 Free PMC article.
-
Autophagy Regulatory Genes MET and RIPK2 Play a Prognostic Role in Pancreatic Ductal Adenocarcinoma: A Bioinformatic Analysis Based on GEO and TCGA.Biomed Res Int. 2020 Nov 5;2020:8537381. doi: 10.1155/2020/8537381. eCollection 2020. Biomed Res Int. 2020. PMID: 33204717 Free PMC article.
-
Identification of P-Rex1 in the Regulation of Liver Cancer Cell Proliferation and Migration via HGF/c-Met/Akt Pathway.Onco Targets Ther. 2020 Sep 24;13:9481-9495. doi: 10.2147/OTT.S265592. eCollection 2020. Onco Targets Ther. 2020. PMID: 33061433 Free PMC article.
-
Lidocaine attenuates TMZ resistance and inhibits cell migration by modulating the MET pathway in glioblastoma cells.Oncol Rep. 2024 May;51(5):72. doi: 10.3892/or.2024.8731. Epub 2024 Apr 12. Oncol Rep. 2024. PMID: 38606513 Free PMC article.
-
Ovarian Tumor Microenvironment Signaling: Convergence on the Rac1 GTPase.Cancers (Basel). 2018 Sep 27;10(10):358. doi: 10.3390/cancers10100358. Cancers (Basel). 2018. PMID: 30261690 Free PMC article. Review.
References
-
- Koochekpour S., Jeffers M., Rulong S., Taylor G., Klineberg E., Hudson E. A., Resau J. H., and Vande Woude G. F. (1997) Met and hepatocyte growth factor/scatter factor expression in human gliomas. Cancer Res. 57, 5391–5398 - PubMed
-
- Engelman J. A., Zejnullahu K., Mitsudomi T., Song Y., Hyland C., Park J. O., Lindeman N., Gale C. M., Zhao X., Christensen J., Kosaka T., Holmes A. J., Rogers A. M., Cappuzzo F., Mok T., et al. (2007) MET amplification leads to gefitinib resistance in lung cancer by activating ERBB3 signaling. Science 316, 1039–1043 10.1126/science.1141478 - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

