Tualang Honey Reduced Neuroinflammation and Caspase-3 Activity in Rat Brain after Kainic Acid-Induced Status Epilepticus
- PMID: 30108663
- PMCID: PMC6077521
- DOI: 10.1155/2018/7287820
Tualang Honey Reduced Neuroinflammation and Caspase-3 Activity in Rat Brain after Kainic Acid-Induced Status Epilepticus
Abstract
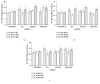
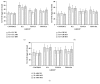
The protective effect of tualang honey (TH) on neuroinflammation and caspase-3 activity in rat cerebral cortex, cerebellum, and brainstem after kainic acid- (KA-) induced status epilepticus was investigated. Male Sprague-Dawley rats were pretreated orally with TH (1.0 g/kg body weight) five times at 12 h intervals. KA (15 mg/kg body weight) was injected subcutaneously 30 min after last oral treatment. Rats were sacrificed at 2 h, 24 h, and 48 h after KA administration. Neuroinflammation markers and caspase-3 activity were analyzed in different brain regions 2 h, 24 h, and 48 h after KA administration. Administration of KA induced epileptic seizures. KA caused significant (p < 0.05) increase in the level of tumor necrosis factor alpha (TNF-α), interleukin 1 beta (IL-1β), glial fibrillary acidic protein (GFAP), allograft inflammatory factor 1 (AIF-1), and cyclooxygenase-2 (COX-2) and increase in the caspase-3 activity in the rat cerebral cortex, cerebellum, and brainstem at multiple time points. Pretreatment with TH significantly (p < 0.05) reduced the elevation of TNF-α, IL-1β, GFAP, AIF-1, and COX-2 level in those brain regions at multiple time points and attenuated the increased caspase-3 activity in the cerebral cortex. In conclusion, TH reduced neuroinflammation and caspase-3 activity after kainic acid- (KA-) induced status epilepticus.
Figures





Similar articles
-
Effect of tualang honey against KA-induced oxidative stress and neurodegeneration in the cortex of rats.BMC Complement Altern Med. 2017 Jan 9;17(1):31. doi: 10.1186/s12906-016-1534-x. BMC Complement Altern Med. 2017. PMID: 28068984 Free PMC article.
-
Propolis ameliorates tumor nerosis factor-α, nitric oxide levels, caspase-3 and nitric oxide synthase activities in kainic acid mediated excitotoxicity in rat brain.Afr J Tradit Complement Altern Med. 2014 Aug 23;11(5):48-53. doi: 10.4314/ajtcam.v11i5.8. eCollection 2014. Afr J Tradit Complement Altern Med. 2014. PMID: 25395704 Free PMC article.
-
Inhibiting HIF-1α Decreases Expression of TNF-α and Caspase-3 in Specific Brain Regions Exposed Kainic Acid-Induced Status Epilepticus.Cell Physiol Biochem. 2016;38(1):75-82. doi: 10.1159/000438610. Epub 2016 Jan 8. Cell Physiol Biochem. 2016. PMID: 26741705
-
Prevention of kainic acid seizures-induced changes in levels of nitric oxide and high-energy phosphates by 7-nitroindazole in rat brain regions.Brain Res. 2003 Aug 15;981(1-2):184-92. doi: 10.1016/s0006-8993(03)03034-8. Brain Res. 2003. PMID: 12885440
-
IL-1β induction and IL-6 suppression are associated with aggravated neuronal damage in a lipopolysaccharide-pretreated kainic acid-induced rat pup seizure model.Neuroimmunomodulation. 2012;19(5):319-25. doi: 10.1159/000339579. Epub 2012 Jul 12. Neuroimmunomodulation. 2012. PMID: 22797174
Cited by
-
Impact of Rapid Eye Movement Sleep Deprivation on Pain Behaviour and Oxidative Stress in the Thalamus: Role of Tualang Honey Supplementation.Malays J Med Sci. 2022 Apr;29(2):69-79. doi: 10.21315/mjms2022.29.2.7. Epub 2022 Apr 21. Malays J Med Sci. 2022. PMID: 35528807 Free PMC article.
-
Natural Products and Their Bioactive Compounds: Neuroprotective Potentials against Neurodegenerative Diseases.Evid Based Complement Alternat Med. 2020 Feb 14;2020:6565396. doi: 10.1155/2020/6565396. eCollection 2020. Evid Based Complement Alternat Med. 2020. PMID: 32148547 Free PMC article. Review.
-
Honey on brain health: A promising brain booster.Front Aging Neurosci. 2023 Jan 17;14:1092596. doi: 10.3389/fnagi.2022.1092596. eCollection 2022. Front Aging Neurosci. 2023. PMID: 36733498 Free PMC article. Review.
-
Elucidating the Multi-Targeted Role of Nutraceuticals: A Complementary Therapy to Starve Neurodegenerative Diseases.Int J Mol Sci. 2021 Apr 14;22(8):4045. doi: 10.3390/ijms22084045. Int J Mol Sci. 2021. PMID: 33919895 Free PMC article. Review.
-
Medicinal activities of Tualang honey: a systematic review.BMC Complement Med Ther. 2024 Oct 4;24(1):358. doi: 10.1186/s12906-024-04664-2. BMC Complement Med Ther. 2024. PMID: 39367403 Free PMC article.
References
-
- Gao H.-M., Zhou H., Hong J.-S. Oxidative stress, neuroinflammation, and neurodegeneration. Neuroinflammation and Neurodegeneration. 2014:81–104. doi: 10.1007/978-1-4939-1071-7. - DOI
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

