Synthesis and antitumor mechanism of a new iron(iii) complex with 5,7-dichloro-2-methyl-8-quinolinol as ligands
- PMID: 30108780
- PMCID: PMC6072324
- DOI: 10.1039/c6md00644b
Synthesis and antitumor mechanism of a new iron(iii) complex with 5,7-dichloro-2-methyl-8-quinolinol as ligands
Abstract
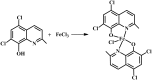
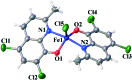
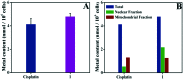
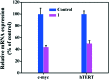
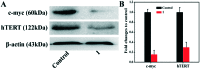
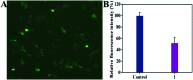
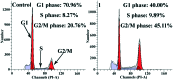
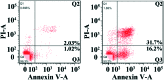
A new iron(iii) complex with 5,7-dichloro-2-methyl-8-quinolinol (HClMQ) as ligands, i.e., [Fe(ClMQ)2Cl] (1), was synthesized and evaluated for its anticancer activity. Compared to the HClMQ ligand, complex 1 showed a higher cytotoxicity towards a series of tumor cell lines, including Hep-G2, BEL-7404, NCI-H460, A549, and T-24, with IC50 values in the range of 5.04-14.35 μM. Notably, the Hep-G2 cell line was the most sensitive to complex 1. Mechanistic studies indicated that complex 1 is a telomerase inhibitor targeting c-myc G-quadruplex DNA and can trigger cell apoptosis via inducing cell cycle arrest and DNA damage.
Figures

References
-
- Maji B., Bhattacharya S. Chem. Commun. 2014;50:6422–6438. - PubMed
-
- Franceschin M., Rizzo A., Casagrande V., Salvati E., Alvino A., Altieri A., Ciammaichella A., Lachettini S., Leonetti C., Ortaggi G., Porru M., Bianco A., Biroccio A. ChemMedChem. 2012;7:2144–2154. - PubMed
-
- Karim N. H. A., Mendoza O., Shivalingam A., Thompson A. J., Ghosh S., Kuimova M. K., Vilar R. RSC Adv. 2014;4:3355–3363.
-
- Muller S., Rodriguez R. Expert. Rev. Clin. Pharmacol. 2014;7:663–679. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources

