Criterion for trismus in head and neck cancer patients: a verification study
- PMID: 30109487
- PMCID: PMC6373229
- DOI: 10.1007/s00520-018-4402-z
Criterion for trismus in head and neck cancer patients: a verification study
Abstract
Purpose: Several cut-off points for trismus in head and neck cancer patients have been used. A mouth opening of 35 mm or less is most frequently used as cut-off point. Due to the variation in cut-off points, prevalence, risk factors and treatment outcomes of trismus cannot be studied in a uniform manner. To provide uniformity, we aimed to verify the cut-off point of 35 mm or less. Additionally, we aimed to determine associated covariates with reported difficulties when opening the mouth.
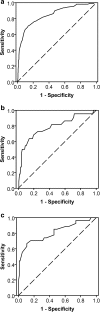
Methods: In a cross-sectional design, we measured the mouth opening in 671 head and neck cancer patients at the Department of Oral and Maxillofacial Surgery, at the University Medical Center Groningen. The cut-off point was determined using the receiver operating characteristic curve and Youden index, with reported difficulties when opening the mouth as criterion for trismus. Cut-off points for significant covariates were also determined.
Results: The Youden index was highest at 35 mm, with a sensitivity of 0.71 and a specificity of 0.86. Of the covariates analysed, type of treatment modality was significantly associated with reported difficulties when opening the mouth. The highest Youden index for patients treated with surgery alone was 37 mm and for patients treated with radiotherapy alone 33 mm.
Conclusions: The cut-off point of 35 mm or less for trismus was confirmed in a head and neck cancer population and is recommended to be used in future studies. Patients receiving different treatment modalities experience difficulty when opening the mouth differently.
Keywords: Head and neck neoplasms; Mouth neoplasms; Quality of life; Range of motion, articular; Surgery, oral; Trismus.
Conflict of interest statement
Research involving human participants and/or animals
Retrospective study: for this type of study formal consent is not required.
The Medical Ethical Committee of the UMCG concluded that our research was not subject to the Medical Research (Human Subject) Act (METc number 2016.692).
This article does not contain any studies with animals performed by any of the authors.
Conflict of interest
None declared.
Data
Corresponding author has full control of all primary data.
Corresponding author allows the journal to review the data if requested.
Figures

References
-
- Bensadoun RJ, Riesenbeck D, Lockhart PB, Elting LS, Spijkervet FK, Brennan MT, Trismus Section, Oral Care Study Group, Multinational Association for Supportive Care in Cancer (MASCC)/International Society of Oral Oncology (ISOO) A systematic review of trismus induced by cancer therapies in head and neck cancer patients. Support Care Cancer. 2010;18:1033–1038. doi: 10.1007/s00520-010-0847-4. - DOI - PubMed
-
- Melchers LJ, Van Weert E, Beurskens CHG, Reintsema H, Slagter AP, Roodenburg JLN, Dijkstra PU. Exercise adherence in patients with trismus due to head and neck oncology: a qualitative study into the use of the Therabite®. Int J Oral Maxillofac Surg. 2009;38:947–954. doi: 10.1016/j.ijom.2009.04.003. - DOI - PubMed
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

