Pilot Study of Immunoblots with Recombinant Borrelia burgdorferi Antigens for Laboratory Diagnosis of Lyme Disease
- PMID: 30110913
- PMCID: PMC6163603
- DOI: 10.3390/healthcare6030099
Pilot Study of Immunoblots with Recombinant Borrelia burgdorferi Antigens for Laboratory Diagnosis of Lyme Disease
Abstract
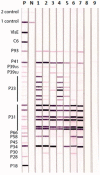
Accurate laboratory diagnosis of Lyme disease (Lyme borreliosis), caused by the spirochete Borrelia burgdorferi (BB), is difficult and yet important to prevent serious disease. The US Centers for Disease Control and Prevention (CDC) presently recommends a screening test for serum antibodies followed by confirmation with a more specific Western blot (WB) test to detect IgG and IgM antibodies against antigens in whole cell lysates of BB. Borrelia species related to BB cause tick-borne relapsing fever (TBRF). TBRF is increasingly recognized as a health problem in the US and occurs in areas where Lyme disease is prevalent. The two groups of Borrelia share related antigens. We have developed a modified WB procedure termed the Lyme immunoblots (IBs) using recombinant antigens from common strains and species of the BB sensu lato complex for serological diagnosis of Lyme disease. A reference collection of 178 sera from 26 with and 152 patients without Lyme disease were assessed by WB and IB in a blinded manner using either criteria for positive antibody reactions recommended by the CDC or criteria developed in-house. The sensitivity, specificity, positive and negative predictive values obtained with the reference sera suggest that the Lyme IB is superior to the Lyme WB for detection of specific antibodies in Lyme disease. The Lyme IB showed no significant reaction with rabbit antisera produced against two Borrelia species causing TBRF in the US, suggesting that the Lyme IB may be also useful for excluding TBRF.
Keywords: Borrelia burgdorferi; Lyme disease; immunoblot; laboratory diagnosis; tick-borne diseases; western blot.
Conflict of interest statement
J.S. and I.D.C. own stock in IGeneX. I.D.C., C.C.R. and P.T. are employees of IGeneX Inc. R.R. and S.L. are affiliated to IGeneX.
Figures


References
LinkOut - more resources
Full Text Sources
Other Literature Sources

